- 1 Explore 3-FPM for Sale – Options, Safety, and Key Insights
- 2 The Growing Appeal of 3-FPM Products
- 3 How to Choose Reliable 3-FPM Vendors
- 4 Enjoy the Convenience of 3-FPM Online Shops
- 5 Tips for Safe and Satisfying 3-FPM Purchases
- 6 Experience the Benefits of 3-FPM
- 7 Summary
- 8 Chemistry
- 9 Pharmacology
- 10 Legality
- 11 FAQ
- 11.1 1. What is 3-Fluorophenmetrazine (3-FPM)?
- 11.2 2. Is 3-FPM legal in the United States?
- 11.3 3. When did 3-FPM become illegal in Virginia, USA?
- 11.4 4. Are other positional isomers of 3-FPM illegal in Virginia or at the federal level in the USA?
- 11.5 5. Is 3-FPM illegal in Sweden?
- 11.6 6. Is 3-FPM legal in Switzerland?
- 12 References
Explore 3-FPM for Sale – Options, Safety, and Key Insights
If you’re in the market for quality 3-FPM products, understanding your options is crucial. With its growing popularity, 3-FPM is highly sought after for research purposes, offering versatility and powerful effects. Whether you want to buy 3-FPM online, explore 3-FPM for sale in the USA or Canada, or connect with reliable 3-FPM vendors, knowing where to shop and how to ensure safety will enhance your buying experience.
The Growing Appeal of 3-FPM Products
3-FPM stands out in the research chemical market for its effectiveness and reliability. Available in various forms, it caters to diverse preferences and applications. With the ability to buy 3-FPM online conveniently through verified platforms, researchers and enthusiasts can access high-quality 3-FPM research chemicals without hassle.
Globally accessible options ensure that you can easily buy 3-FPM USA, 3-FPM Canada, or even explore international shipping. Trusted 3-FPM vendors provide a range of products, including bulk purchase options for those requiring large quantities. Discreet shipping and secure payment methods further enhance the appeal, making the process smooth and safe.
How to Choose Reliable 3-FPM Vendors
Purchasing chemicals like 3-FPM requires careful consideration of the vendor’s reputation and transparency. Here’s what to keep in mind:
- Check Vendor Reviews
Look for vendors with positive feedback on their 3-FPM online platforms. Reputation is everything in this market.
- Detail-Oriented Listings
Trusted suppliers provide clear and detailed descriptions of their 3-FPM research chemicals, helping you understand product quality and structure.
- Global Shipping and Accessibility
A vendor catering to diverse regions and enabling purchases like order 3-FPM USA or buy 3-FPM Australia ensures accessibility wherever you are.
Enjoy the Convenience of 3-FPM Online Shops
The ability to purchase 3-FPM from the comfort of your home makes online platforms increasingly popular. Whether you’re browsing 3-FPM shop options for personal research or looking to purchase specific quantities, online stores simplify the process by offering a wide selection and competitive pricing. You can find everything from 3-FPM powder to bulk quantities with just a few clicks.
Opt for vendors who prioritize security and discretion, especially when placing an order 3-FPM online. Platforms offering guidance, responsive customer service, and secure payment systems provide peace of mind for every transaction.
Tips for Safe and Satisfying 3-FPM Purchases
- Verify Product Quality
Always ensure that the vendor offers lab-tested 3-FPM research chemicals with proven purity.
- Understand Pricing and Shipping Costs
Compare pricing across trusted platforms. Terms like 3-FPM sale often indicate discounts, but verify the total cost, including shipping.
- Choose Discreet and Reliable Shipping
Vendors specializing in buy 3-FPM online USA or international shipping should prioritize discreet packaging to protect your privacy.
By following these steps, you can confidently explore options for 3-FPM for sale and enjoy a smooth purchasing experience.
Experience the Benefits of 3-FPM
Whether you’re a researcher or an enthusiast, sourcing high-quality products like 3-FPM powder has never been easier. Reputable vendors simplify the process, enabling you to purchase 3-FPM safely and confidently. From 3-FPM buy online USA options to finding trusted suppliers globally, the thriving market ensures there’s something for every need.
Call to Action
Explore top-rated vendors for 3-FPM and secure high-quality products today. Whether you’re based in the USA, Canada, Australia, or beyond, find trusted 3-FPM shop options that prioritize safety, quality, and convenience. Start your online shopping experience now!
Summary
3-Fluorophenmetrazine, commonly referred to as 3-FPM and also known as 3-FPH and PAL-593, is a stimulant with its foundation in phenylmorpholine. This chemical compound serves as a fluorinated derivative of phenmetrazine and has been distributed via online sources under the guise of a designer drug.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1350768-28-3 HCl: 1803562-83-5 |
|---|---|
| PubChem CID | 54673723HCl: 75481405 |
| ChemSpider | 37509994HCl: 34212559 |
| UNII | BEV6RF569G |
| Chemical and physical data | |
| Formula | C11H14FNO |
| Molar mass | 195.237 g·mol−1 |
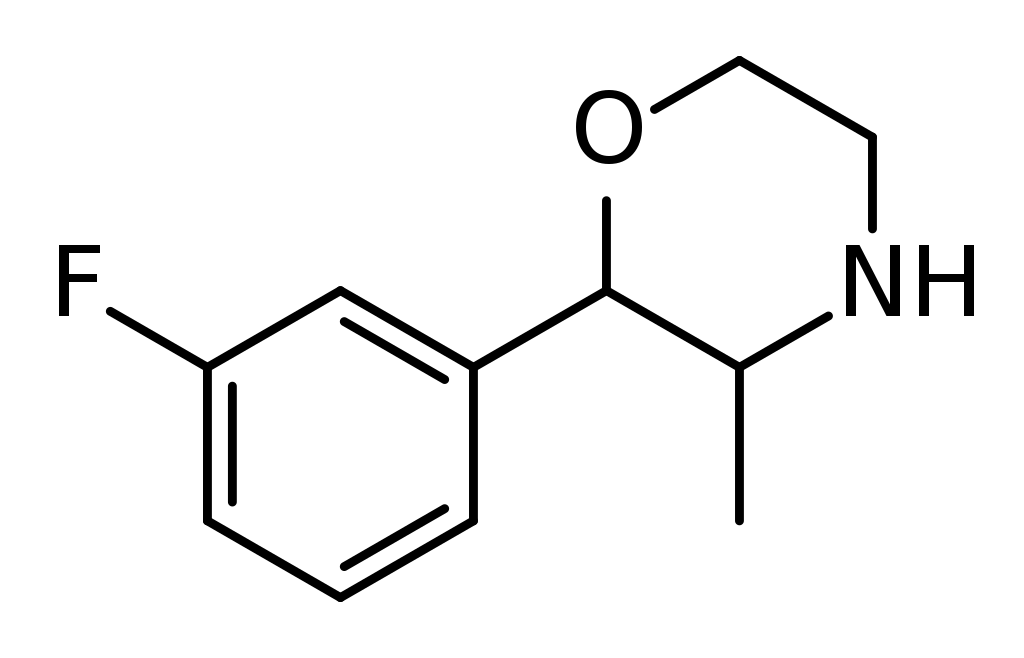
Chemistry
3-Fluorophenmetrazine, belonging to the morpholine class, is a fluorinated counterpart of phenmetrazine, a stimulant.
It’s worth noting that 3-Fluorophenmetrazine is a regioisomer, differing in chemical structure, from both 2-fluorophenmetrazine and 4-fluorophenmetrazine.
Pharmacology
3-FPM exhibits its actions as a norepinephrine–dopamine releasing agent, with EC50 values of 30 nM and 43 nM, respectively, indicating its potency in releasing these neurotransmitters. However, it displays minimal effectiveness in promoting serotonin release, as evidenced by an EC50 value of 2558 nM.
Additionally, 3-FPM hinders the uptake facilitated by dopamine transporters and norepinephrine transporters in HEK293 cells, showing potencies similar to cocaine, with IC50 values less than 2.5 μM. However, its impact on serotonin transporters is less pronounced, with IC50 values exceeding 80 μM.
At sufficiently high doses, 3-FPM can reverse the action of monoamine transporters, particularly those associated with the catecholamines dopamine and norepinephrine, and, to a lesser extent, serotonin transporters. This reversal leads to the release of these neurotransmitters from the cytosol into the extracellular space, where they become active.
Studies into its metabolic pathway have revealed processes such as N-oxidation, aryl hydroxylation followed by O-methylation, alkyl hydroxylation, oxidation, and the degradation of the ethyl-bridge, resulting in the formation of the O/N-bis-dealkylated metabolite. These processes can occur in combination with further glucuronidation or sulfation reactions.
Legality
In the United States, 3-fluorophenmetrazine is not explicitly prohibited at the federal level. However, it could potentially fall under the federal Analogue Act if it is intended for human consumption and is considered a structural analog of the Schedule II drug Phenmetrazine.
On November 16, 2016, 3-fluorophenmetrazine was banned in the state of Virginia. As of 2019, it is classified as a Schedule I substance in Virginia. Similarly, the positional isomers of 3-fluorophenmetrazine, such as 2-fluorophenmetrazine and 4-fluorophenmetrazine, are also illegal under Virginia state law but not under federal law.
Sweden’s public health agency proposed the classification of 3-Fluorophenmetrazine as an illegal narcotic on June 1, 2015. It was officially classified as such on October 15, 2015.
Furthermore, 3-fluorophenmetrazine has been illegal in Switzerland since December 2015.
FAQ
1. What is 3-Fluorophenmetrazine (3-FPM)?
- 3-Fluorophenmetrazine, often referred to as 3-FPM, is a chemical compound that belongs to the phenylmorpholine class. It is known for its stimulant properties and is considered a derivative of Phenmetrazine.
2. Is 3-FPM legal in the United States?
- At the federal level, 3-FPM is not explicitly illegal. However, it may be subject to regulation under the federal Analogue Act if it is intended for human consumption and considered a structural analog of the Schedule II drug Phenmetrazine.
3. When did 3-FPM become illegal in Virginia, USA?
- 3-FPM was classified as an illegal substance in the state of Virginia on November 16, 2016. As of 2019, it is categorized as a Schedule I substance in Virginia.
4. Are other positional isomers of 3-FPM illegal in Virginia or at the federal level in the USA?
- Yes, other positional isomers of 3-FPM, such as 2-fluorophenmetrazine and 4-fluorophenmetrazine, are also illegal under Virginia state law. However, they are not prohibited under federal law.
5. Is 3-FPM illegal in Sweden?
- Yes, Sweden’s public health agency proposed classifying 3-fluorophenmetrazine as an illegal narcotic on June 1, 2015. It was officially classified as such on October 15, 2015.
6. Is 3-FPM legal in Switzerland?
- No, 3-Fluorophenmetrazine is illegal in Switzerland as of December 2015.
Please note that legal status and regulations regarding 3-Fluorophenmetrazine may vary by country and region, and it is important to be aware of and comply with local laws and regulations regarding its use and possession. Additionally, its use should always be approached with caution due to its potential health risks and unknown long-term effects.
References
- McLaughlin G, Morris N, Kavanagh PV, Dowling G, Power JD, Twamley B, et al. (March 2017). “Test purchase, synthesis and characterization of 3-fluorophenmetrazine (3-FPM) and differentiation from its ortho- and para-substituted isomers” (PDF). Drug Testing and Analysis. 9 (3): 369–377. doi:10.1002/dta.1945. PMID 26810957. S2CID 205762700.
- Bäckberg M, Westerbergh J, Beck O, Helander A (November 2016). “Adverse events related to the new psychoactive substance 3-fluorophenmetrazine – results from the Swedish STRIDA project”. Clinical Toxicology. 54 (9): 819–825. doi:10.1080/15563650.2016.1211288. PMID 27491700. S2CID 26118285.
- Bruce E. Blough; Richard Rothman; Antonio Landavazo; Kevin M. Page; Ann Marie Decker (8 August 2013). “US Patent 20130203752 A1 – Phenylmorpholines and analogues thereof”.
- Mayer FP, Burchardt NV, Decker AM, Partilla JS, Li Y, McLaughlin G, et al. (May 2018). “Fluorinated phenmetrazine “legal highs” act as substrates for high-affinity monoamine transporters of the SLC6 family”. Neuropharmacology. 134 (Pt A): 149–157. doi:10.1016/j.neuropharm.2017.10.006. PMC 7294773. PMID 28988906.
- Mardal M, Miserez B, Bade R, Portolés T, Bischoff M, Hernández F, Meyer MR (September 2016). “3-Fluorophenmetrazine, a fluorinated analogue of phenmetrazine: Studies on in vivo metabolism in rat and human, in vitro metabolism in human CYP isoenzymes and microbial biotransformation in Pseudomonas Putida and wastewater using GC and LC coupled to (HR)-MS techniques”. Journal of Pharmaceutical and Biomedical Analysis. 128: 485–495. doi:10.1016/j.jpba.2016.06.011. PMID 27372653.
- “VA.R. Doc. No. R17-4746”. 17 December 2018.
- “§ 54.1-3446. Schedule I.” Retrieved 8 September 2023.
- “21 U.S. Code § 812 – Schedules of controlled substances”. 8 September 2023.
- “23 nya ämnen kan klassas som narkotika eller hälsofarlig vara” (in Swedish). Folkhälsomyndigheten. 1 June 2015.
- “Nya substanser klassas som narkotika eller hälsofarlig vara” (in Swedish). Folkhälsomyndigheten. 18 August 2015.
- “Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien” (in German). Der Bundesrat.
