Beautiful Plants For Your Interior
Summary
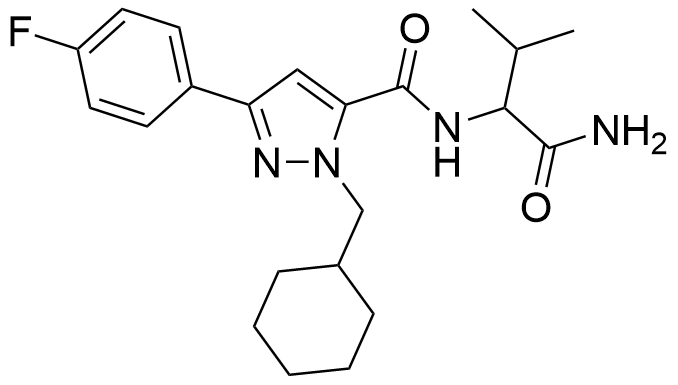
AB-CHFUPYCA, also known as AB-CHMFUPPYCA, is a compound initially discovered as part of synthetic cannabis products in Japan back in 2015. The name “AB-CHFUPYCA” is an abbreviation derived from its systematic name, N-(1-Amino-3-methyl-1-oxoButan-2-yl)-1-(cyclohexylmethyl)-3-(4-FlUorophenyl)-1H-PYrazole-5-CarboxAmide. Two distinct regioisomers of AB-CHFUPYCA exist: 3,5-AB-CHMFUPPYCA and 5,3-AB-CHMFUPPYCA. Both collectively refer to as AB-CHMFUPPYCA isomers, meaning that AB-CHMFUPPYCA and AB-CHFUPYCA don’t represent unique chemical structures.
AB-CHFUPYCA shares structural similarities with other synthetic cannabinoids like 5F-AB-FUPPYCA, JWH-307, JWH-030, JWH-147, and AB-PINACA. It could be considered an analog of the traditional pyrazole cannabinoid receptor one antagonist, rimonabant. Notably, the specific pharmacological properties of AB-CHFUPYCA have yet to be subject to extensive study.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1870799-79-3 |
|---|---|
| PubChem CID | 122213879 |
| ChemSpider | 67168777 |
| UNII | O4PS4BDB8K |
| Chemical and physical data | |
| Formula | C22H29FN4O2 |
| Molar mass | 400.498 g·mol−1 |
FAQ
1. What is AB-CHFUPYCA, and where was it initially identified?
AB-CHFUPYCA, also known as AB-CHMFUPPYCA, is a chemical compound first discovered in synthetic cannabis products in Japan in 2015.
2. What does the name AB-CHFUPYCA stand for?
The name “AB-CHFUPYCA” is an acronym derived from its systematic name, N-(1-Amino-3-methyl-1-oxoButan-2-yl)-1-(cyclohexylmethyl)-3-(4-FlUorophenyl)-1H-PYrazole-5-CarboxAmide.
3. How many regioisomers of AB-CHFUPYCA are known to exist, and what are they called?
There are two recognized regioisomers of AB-CHFUPYCA: 3,5-AB-CHMFUPPYCA and 5,3-AB-CHMFUPPYCA. Both are collectively referred to as AB-CHMFUPPYCA isomers.
4. Are AB-CHFUPYCA and AB-CHMFUPPYCA unique chemical structures?
No, AB-CHFUPYCA and AB-CHMFUPPYCA do not represent distinct chemical structures. They are names for the same compound with different regioisomers.
5. How does AB-CHFUPYCA relate to other synthetic cannabinoids?
AB-CHFUPYCA shares structural elements with other synthetic cannabinoids, including 5F-AB-FUPPYCA, JWH-307, JWH-030, JWH-147, and AB-PINACA. It can be considered an analog of the traditional pyrazole cannabinoid receptor one antagonist, rimonabant.
6. Have the pharmacological properties of AB-CHFUPYCA been studied?
As of now, there is limited information available about the pharmacological properties of AB-CHFUPYCA. Further research is needed to understand its effects and potential risks better.
7. Is AB-CHFUPYCA subject to legal regulations?
The legal status of AB-CHFUPYCA may vary by region. It is important to know local laws and restrictions on its use and distribution.
8. Are there any health concerns associated with AB-CHFUPYCA use?
Due to limited research, potential health risks, and variations in legal status, it is advisable to exercise caution and seek guidance from healthcare professionals and regulatory authorities when dealing with substances like AB-CHFUPYCA.
References
- McLaughlin G, Morris N, Kavanagh PV, Power JD, Twamley B, O’Brien J, et al. (September 2016). “The synthesis and characterization of the ‘research chemical’ 3,5-AB-CHMFUPPYCA”: This study focuses on the synthesis and characterization of 3,5-AB-CHMFUPPYCA, a synthetic cannabinoid, and distinguishes it from its regioisomer, providing valuable insights into its chemical properties.
- Uchiyama N, Asakawa K, Kikura-Hanajiri R, Tsutsumi T, Hakamatsuka T (August 2015). “Identification of AB-CHFUPYCA in illegal products”: This research identifies AB-CHFUPYCA in illicit products, shedding light on its presence in the black market and potential regulatory concerns.
- Franz F, Angerer V, Brandt SD, McLaughlin G, Kavanagh PV, Moosmann B, Auwärter V (February 2017). “In vitro metabolism of 3,5-AB-CHMFUPPYCA”: This study delves into the metabolism of 3,5-AB-CHMFUPPYCA and its regioisomer, providing insights into how the body processes these synthetic cannabinoids and their potential effects.
- “5-fluoro-3,5-AB-PFUPPYCA” – Cayman Chemical Company: Cayman Chemical offers information about 5-fluoro-3,5-AB-PFUPPYCA, a related compound, providing valuable data for researchers and individuals interested in these synthetic substances.