Beautiful Plants For Your Interior
Summary
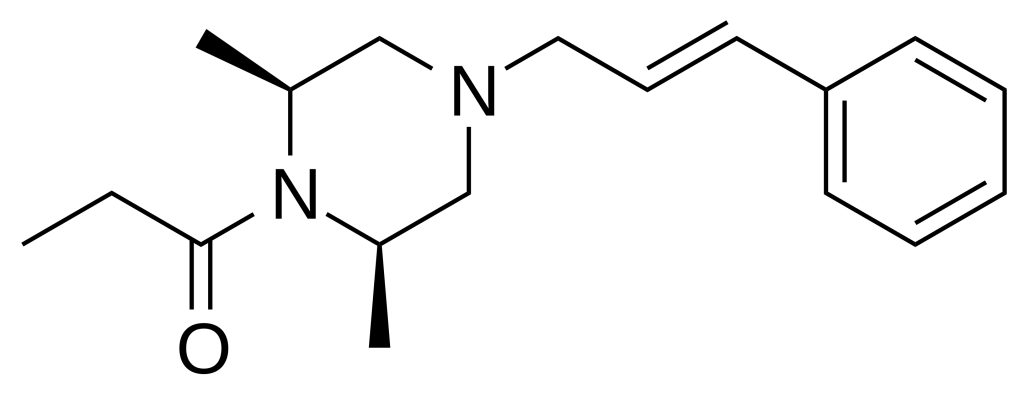
AP-238 is an opioid-designer drug sharing chemical kinship with compounds like azaprocin and bucinnazine, exhibiting a potency level akin to that of morphine. Originating in Italy during the 1960s, it remained unmarketed yet emerged on the underground market approximately in 2020, making notable appearances in both Slovenia and the United States.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | (racemic) 4204-01-7 (racemic) 140924-11-4 (2R,6S enantiomer) |
|---|---|
| PubChem CID | 124389619 |
| ChemSpider | 4941886 |
| UNII | N26WML8AR7 |
| Chemical and physical data | |
| Formula | C18H26N2O |
| Molar mass | 286.419 g·mol−1 |
FAQ
- What is AP-238?
- AP-238 is an opioid-designer drug that is chemically related to substances like azaprocin and bucinnazine. It possesses a potency similar to that of morphine.
- When was AP-238 discovered?
- AP-238 was initially discovered in Italy during the 1960s. However, it was never introduced into the legal pharmaceutical market.
- When did AP-238 appear on the illicit market?
- AP-238 made its presence felt on the illicit market around 2020.
- In which countries have AP-238 been detected?
- AP-238 has been detected in both Slovenia and the United States.
- Is AP-238 legal to possess or use?
- The legality of AP-238 varies by country and region. It is essential to be aware of the specific regulations in your area regarding the possession and use of this substance.
- What are the effects of AP-238?
- As an opioid, AP-238 may produce effects similar to other opioids, including pain relief and feelings of euphoria. However, it also carries the risk of side effects and potential addiction.
- Is AP-238 safe to use?
- The safety of AP-238 is not well-documented, and it may carry health risks, including the risk of overdose and addiction. Using such substances can be dangerous and should be avoided.
- Is AP-238 for medical use?
- AP-238 is not approved for medical use and is primarily considered a designer or recreational drug.
- Are there any known health risks associated with AP-238?
- The use of AP-238 and similar substances can lead to health risks such as respiratory depression, overdose, and addiction. It is essential to exercise caution and seek professional advice if you have concerns about your health or substance use.
- Where can I find more information about AP-238?
- For comprehensive and up-to-date information on AP-238 and related substances, it is advisable to consult with reputable sources such as government health agencies, poison control centers, or addiction treatment services. Always prioritize your health and well-being when considering the use of any substances.
References
- U.S. Drug Enforcement Administration and 2-Methyl-AP-237The U.S. Drug Enforcement Administration (DEA) provides information about 2-Methyl-AP-237, a synthetic opioid. The agency plays a crucial role in monitoring and regulating controlled substances.
- Toxicological and Pharmacological Research on Novel Cinnamylpiperazine Synthetic OpioidsA study published in June 2022 conducted toxicological and pharmacological research on novel synthetic opioids, including 2-Methyl AP-237 and AP-238. This research aimed to better understand the effects and risks associated with these substances.
- Structurally Related CompoundsIn a May 1968 publication, researchers explored structurally related compounds, including 1-Propionyl-4-substituted cis-2,6-dimethylpiperazines. These compounds are associated with analgesic properties and are relevant to the study of synthetic opioids.
- Analytical Report on AP-238An analytical report from the Nacionalni Forenzični Laboratorij provides insights into AP-238, describing it as “1‐{2,6‐dimethyl‐4‐[(2E)‐3‐phenylprop‐2‐en‐1‐yl]piperazin‐1‐yl}propan‐1‐one.” This information contributes to our understanding of the compound’s chemical structure.
- NPS Discovery at CFSRE and AP-238The Center for Forensic Science Research and Education (CFSRE) published a report in November 2020, discussing AP-238 and its status as a new psychoactive substance (NPS). This report is a valuable resource for those seeking information on AP-238.