Beautiful Plants For Your Interior
Summary
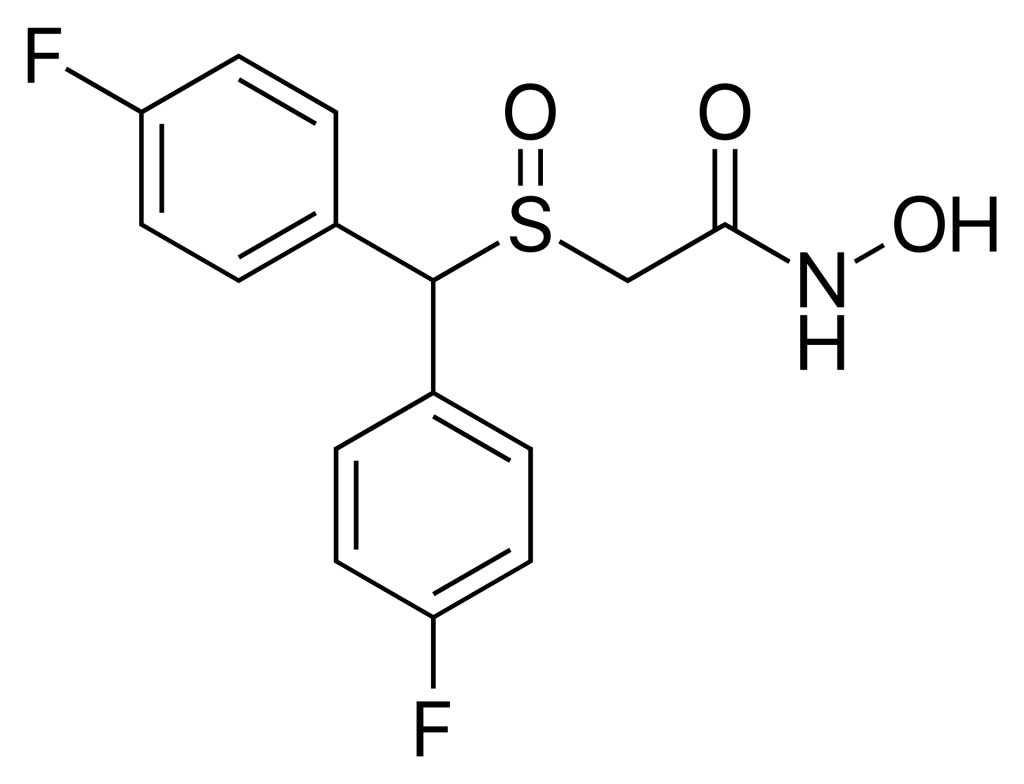
CRL-40,941, known by its alternative names fladrafinil and fluorafinil, falls within the category of eugeroics and shares a close chemical relationship with adrafinil and modafinil. Specifically, it is the bis(p-fluoro) ring-substituted derivative of adrafinil. One noteworthy distinction is that CRL-40,941 has demonstrated anti-aggressive effects in animals, a characteristic not observed with adrafinil, and it is reported to be approximately 3 to 4 times more potent than adrafinil.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 90212-80-9 |
|---|---|
| PubChem CID | 13316557 |
| ChemSpider | 29785194 |
| UNII | 5RT6X0M01F |
| CompTox Dashboard (EPA) | DTXSID40536590 |
| Chemical and physical data | |
| Formula | C15H13F2NO3S |
| Molar mass | 325.33 g·mol−1 |
FAQ
- What is CRL-40,941?
- CRL-40,941, or fladrafinil and fluorafinil, is a chemical compound categorized as a eugeroic. It shares a close chemical relationship with adrafinil and modafinil.
- What is a eugeroic?
- A eugeroic is a class of drugs that promotes wakefulness and alertness. They are often used to address sleep-related disorders and enhance cognitive function.
- How does CRL-40,941 differ from adrafinil and modafinil?
- CRL-40,941 is the bis(p-fluoro) ring-substituted derivative of adrafinil. Notably, it has been found to exhibit anti-aggressive effects in animals, a quality not observed with adrafinil. Additionally, it is purported to be significantly more potent, approximately 3 to 4 times stronger than adrafinil.
- What are the potential applications of CRL-40,941?
- CRL-40,941 is primarily explored for its potential to promote wakefulness and cognitive enhancement. It may be used for sleep disorders or situations requiring heightened alertness.
- Is CRL-40,941 approved for medical use?
- As my last knowledge update in September 2021, CRL-40,941 had not received regulatory approval for medical use in many countries. It is crucial to stay informed about its regulatory status, which may change over time.
- What are the reported side effects of CRL-40,941?
- The side effects of CRL-40,941, particularly with prolonged use, still need to be fully understood. Users should exercise caution and consult healthcare professionals if considering its use.
- Where can I find more information about CRL-40,941?
- To access in-depth information about CRL-40,941, consider referring to scientific literature, research studies, and reliable sources. It’s essential to stay updated on developments regarding its regulatory status and any potential medical applications.
References
- US 4489095, Lafon L, “Halogenobenzhydrylsulfinylacetohydroxamic acids”, published 1984-12-18, assigned to Laboratoire L. Lafon