Beautiful Plants For Your Interior
Summary
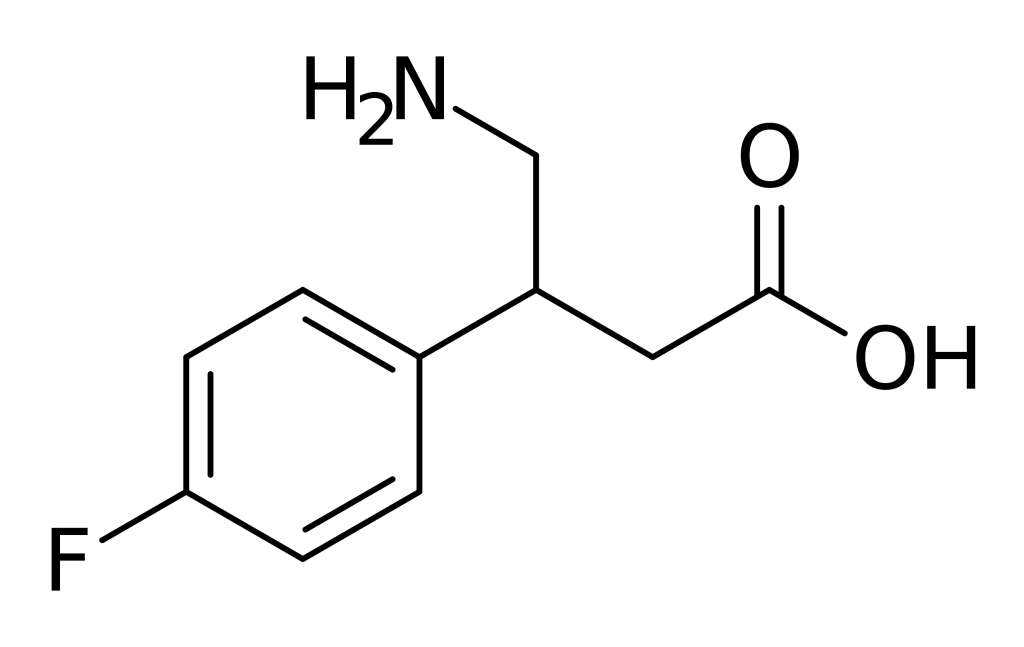
4-Fluorophenibut, which also goes by the developmental code name CGP-11130, is recognized as β-(4-fluorophenyl)-γ-aminobutyric acid or β-(4-fluorophenyl)-GABA. It operates as an agonist for the GABAB receptor, although it never reached the commercial market.
In its selectivity, 4-Fluorophenibut demonstrates a preference for the GABAB receptor over the GABAA receptor, with respective IC50 values of 1.70 μM and >100 μM. Structurally, this compound is a GABAanalogue and shares a close kinship with substances like baclofen (β-(4-chlorophenyl)-GABA), halibut (β-(4-methyl phenyl)-GABA), and halibut (β-phenyl-GABA).
Notably, while 4-fluorophenibut is less potent as a GABAB receptor agonist when compared to baclofen, it exhibits greater potency than phenibut in this regard.
Informally, this substance is sometimes referred to as 4F-phenibut, F-phenibut, or simply as fluorobut.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 52237-19-1 |
|---|---|
| PubChem CID | 103611 |
| ChemSpider | 93547 |
| UNII | 6DAU7M5P3D |
| Chemical and physical data | |
| Formula | C10H12FNO2 |
| Molar mass | 197.209 g·mol−1 |
FAQ
- What is 4-Fluorophenibut?
- 4-Fluorophenibut, also known as CGP-11130, is a chemical compound known as β-(4-fluorophenyl)-γ-aminobutyric acid. It functions as an agonist for the GABAB receptor.
- What is the GABAB receptor, and how does 4-Fluorophenibut interact with it?
- The GABAB receptor is a neurotransmitter receptor in the brain. 4-Fluorophenibut selectively activates the GABAB receptor, affecting neurotransmission.
- Was 4-Fluorophenibut ever commercially available?
- No, 4-Fluorophenibut was never marketed for commercial use.
- How does 4-fluorophenibut differ from other substances like baclofen and phenibut?
- 4-Fluorophenibut is structurally related to baclofen (β-(4-chlorophenyl)-GABA) and phenibut (β-phenyl-GABA). It is less potent as a GABAB receptor agonist compared to baclofen but more potent than phenibut.
- What are some colloquial names for 4-Fluorophenibut?
- Informally, 4-Fluorophenibut is sometimes referred to as 4F-phenibut, F-phenibut, or simply as fluorobut.
- What is the significance of 4-Fluorophenibut’s selectivity for the GABAB receptor over the GABAA receptor?
- This selectivity plays a role in its specific pharmacological effects and potential uses.
- Is 4-Fluorophenibut safe for consumption or recreational use?
- 4-Fluorophenibut’s safety depends on various factors, including its intended use, dosage, and individual tolerance. It’s essential to exercise caution and seek professional advice when considering its use.
- Where can I find more information about 4-Fluorophenibut and its properties?
- You can explore scientific publications, research studies, and academic resources related to chemistry and pharmacology to gain a deeper understanding of 4-Fluorophenibut.
References
- In a study from 1983, Bowery NG, Hill DR, and Hudson AL explored the “Characteristics of GABAB receptor binding sites on rat whole brain synaptic membranes.” Their research was published in the British Journal of Pharmacology, Volume 78, Issue 1, spanning pages 191–206. This work offers valuable insights into GABAB receptor binding. You can access it through DOI: 10.1111/j.1476-5381.1983.tb09380.x and find it in the PMC under the reference PMC 2044790. The PMID is 6297646.
- On the Lithuanian website ntakd.lrv.lt, you’ll discover information titled “RINKOS RIBOJIMO PRIEMONĖS FENIBUTUI!” (in Lithuanian). This resource, retrieved on 2020-01-27, provides details about market control measures for fenibut, a substance related to GABAB receptors.
- For official information from Hungary, you can refer to “MAGYARORSZÁG HIVATALOS LAPJA” (Hungary’s Official Gazette). Retrieved on 2021-04-28, this source is an authoritative reference for legal and official matters in Hungary.