Beautiful Plants For Your Interior
Summary
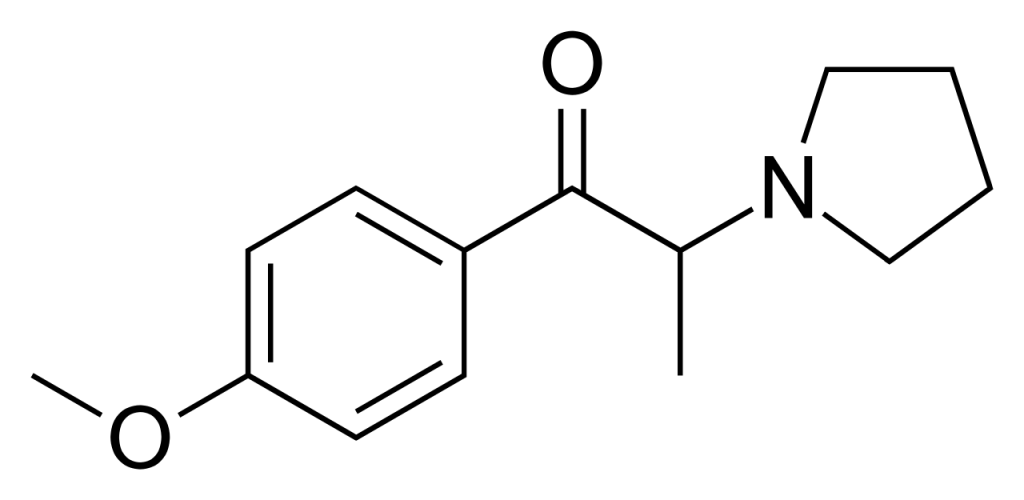
4′-Methoxy-α-pyrrolidinopropiophenone (MOPPP) belongs to the pyrrolidinophenone class of designer stimulant drugs. It can induce euphoria, an effect commonly observed in other traditional stimulants. Please note that additional research and references are needed to verify this claim.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 478243-09-3 |
|---|---|
| PubChem CID | 6430836 |
| ChemSpider | 4936174 |
| UNII | OI15TPX8QP |
| Chemical and physical data | |
| Formula | C14H19NO2 |
| Molar mass | 233.311 g·mol−1 |
Recreational use
MOPPP is relatively uncommonly used compared to other recreational amphetamines or stimulants like meth, cocaine, or speed. It emerged as a designer drug in Germany during the late 1990s and early 2000s, alongside several other derivatives. However, unlike some pyrrolidinophenone counterparts such as α-PPP and MDPV, MOPPP never achieved widespread international popularity.
Recently, there has been a disturbing trend of illicitly selling stimulants under deceptive labels (e.g., as “bath salts”), often containing MDPV as the primary ingredient. Surprisingly, this hasn’t been observed with MOPPP, although it shares a similar potential for abuse.
Chemistry
MOPPP shares a structural similarity with α-PPP much like how PMA is akin to amphetamine. Specifically, MOPPP features the addition of a methoxy group at the 4-position on the phenyl ring, mirroring the structural alteration seen in these analogs.
FAQ
- What is 4′-Methoxy-α-pyrrolidinopropiophenone (MOPPP)?
- MOPPP is a synthetic designer drug belonging to the pyrrolidinophenone class, known for its stimulant properties.
- How is MOPPP Structurally Related to Other Compounds?
- MOPPP is structurally related to α-PPP, similar to how PMA is related to amphetamine. It includes adding a methoxy group at the 4-position on the phenyl ring.
- What Effects Does MOPPP Produce?
- MOPPP can potentially induce euphoria, a sensation commonly associated with classical stimulants. However, further research and references are necessary to confirm its effects.
- Is MOPPP a Commonly Used Recreational Drug?
- No, MOPPP is not commonly used compared to other recreational amphetamines or stimulants like meth, cocaine, or speed.
- When Did MOPPP First Emerge as a Designer Drug?
- MOPPP first emerged as a designer drug in Germany during the late 1990s and early 2000s, alongside several other derivatives.
- Did MOPPP Gain International Popularity Like Some Other Designer Drugs?
- MOPPP did not gain widespread international popularity, unlike other drugs in the pyrrolidinophenone family, such as α-PPP and MDPV.
- Has MOPPP Been Used Deceptively in Products Like “Bath Salts”?
- Unlike some other similar substances, MOPPP has not been observed to be used deceptively in products labelled as “bath salts” despite its potential for abuse.
Please note that information about MOPPP is subject to change as new research and developments occur, and its use may be associated with legal and health risks. It is essential to stay informed and make responsible choices regarding substance use.
References
- In August 2003, Springer D, Fritschi G, and Maurer HH conducted a study titled “Metabolism and toxicological detection of the new designer drug 4′-methoxy-alpha-pyrrolidinopropiophenone studied in rat urine using gas chromatography-mass spectrometry.” This research was published in the Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences, volume 793 (2), pages 331–42. The study aimed to investigate the metabolism and toxicological detection of 4′-methoxy-alpha-pyrrolidinopropiophenone (MOPPP) using rat urine samples.
- DOI: 10.1016/S1570-0232(03)00334-9
- PMID: 12906908
- In October 2003, Springer D, Staack RF, Paul LD, Kraemer T, and Maurer HH conducted a study titled “Identification of cytochrome P450 enzymes involved in the metabolism of 4′-methoxy-alpha-pyrrolidinopropiophenone (MOPPP), a designer drug, in human liver microsomes.” This research was published in Xenobiotica; the Fate of Foreign Compounds in Biological Systems, volume 33 (10), pages 989–98. The study aimed to identify the cytochrome P450 enzymes responsible for metabolizing MOPPP in human liver microsomes.
- DOI: 10.1080/00498250310001602775
- PMID: 14555336
- S2CID: 7838464
- In November 2003, Springer D, Fritschi G, and Maurer HH conducted a study titled “Metabolism of the new designer drug alpha-pyrrolidinopropiophenone (PPP) and the toxicological detection of PPP and 4′-methyl-alpha-pyrrolidinopropiophenone (MPPP) studied in rat urine using gas chromatography-mass spectrometry.” This research was published in the Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences, volume 796 (2), pages 253–66. The study aimed to investigate the metabolism of alpha-pyrrolidinopropiophenone (PPP) and the toxicological detection of PPP and 4′-methyl-alpha-pyrrolidinopropiophenone (MPPP) using rat urine samples.
- DOI: 10.1016/j.jchromb.2003.07.008
- PMID: 14581066
- In April 2004, Maurer HH, Kraemer T, Springer D, and Staack RF authored a synopsis titled “Chemistry, pharmacology, toxicology, and hepatic metabolism of designer drugs of the amphetamine (ecstasy), piperazine, and pyrrolidinophenone types.” This synopsis was published in Therapeutic Drug Monitoring, volume 26 (2), pages 127–31. It provided an overview of the chemistry, pharmacology, toxicology, and hepatic metabolism of designer drugs in the amphetamine, piperazine, and pyrrolidinophenone categories.
- DOI: 10.1097/00007691-200404000-00007
- PMID: 15228152
- S2CID: 9255084
- In June 2005, Staack RF and Maurer HH authored a paper titled “Metabolism of designer drugs of abuse,” which was published in Current Drug Metabolism, volume 6 (3), pages 259–74. This paper discussed the metabolism of designer drugs commonly abused.
- DOI: 10.2174/1389200054021825
- PMID: 15975043
- On March 8, 2011, an article titled “MDPV Bath Salts Drug Over The Counter” was published on Healthbodydaily.com. This article discussed the availability of the drug MDPV, often sold as “bath salts,” without a prescription.
- Source: Healthbodydaily.com (Archived from the original on March 10, 2011)