Beautiful Plants For Your Interior
summary
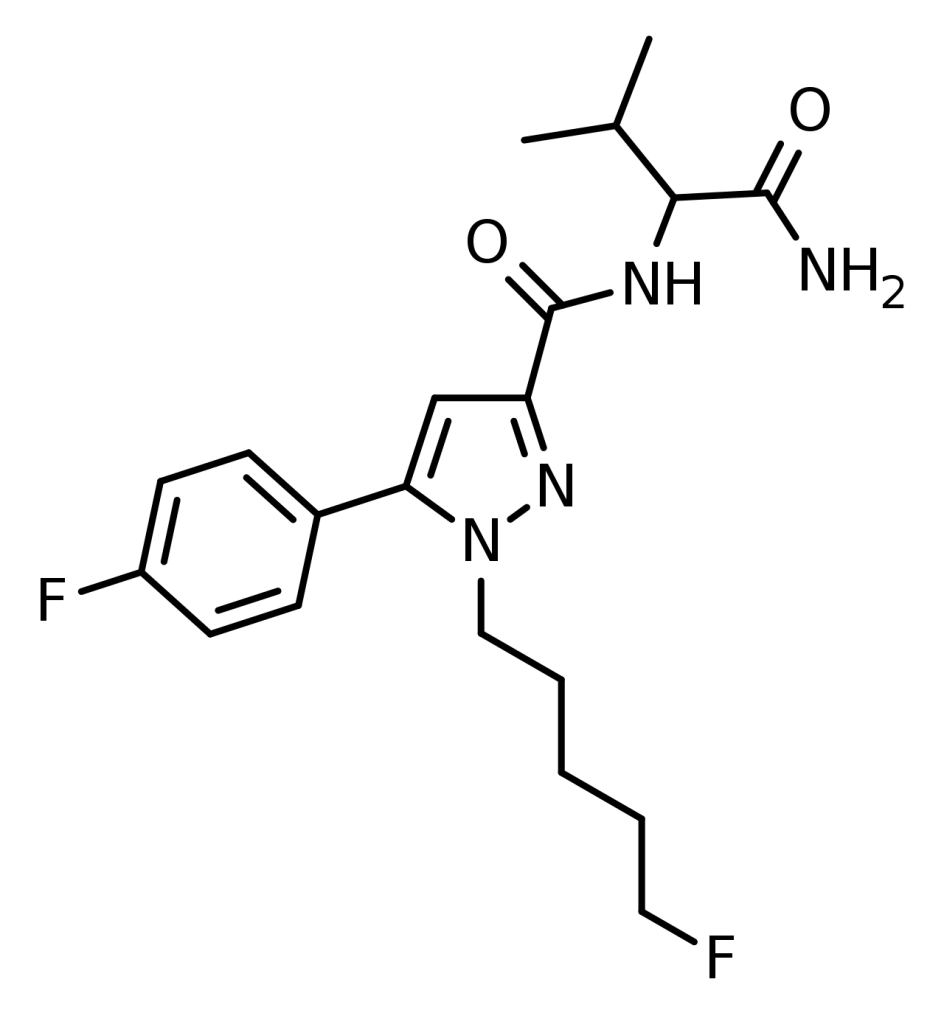
5F-AB-FUPPYCA, or AZ-037, is a synthetic cannabinoid rooted in the pyrazole structure. It is presumed to act as an agonist targeting the CB1 receptor and has gained popularity as a designer drug available for purchase online. Notably, it came into the spotlight when the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) identified it in a seizure of 540 grams of white powder in France in February 2015.
Interestingly, AZ-037 is also a synonym for another compound, 5-fluoro-3,5-AB-PFUPPYCA, which shares a structural isomer with AZ-037. This results in AZ-037 being used interchangeably for these two different substances.
Notably, 5-fluoro-3,5-AB-PFUPPYCA made its appearance in synthetic cannabinoid smoke blends in the United States as early as December 30, 2021, alongside substances like ADB-BUTINACA, MDA-19 (BZO-HEXOXIZID), and MDMB-4en-PINACA.
With structural elements reminiscent of other synthetic cannabinoids, such as AB-CHFUPYCA, JWH-307, JWH-030, JWH-147, and AB-PINACA, 5-fluoro-AB-PFUPPYCA can be considered an analog of the traditional pyrazole cannabinoid receptor one antagonist, rimonabant. However, it’s important to note that the pharmacological properties of 5F-AB-FUPPYCA still need to be explored and unstudied.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 2044702-46-5 |
|---|---|
| PubChem CID | 129317827 |
| ChemSpider | 58191440 |
| UNII | 2G6G797F5B |
| Chemical and physical data | |
| Formula | C20H26F2N4O2 |
| Molar mass | 392.451 g·mol−1 |
Legality
In the United States, as of May 22, 2023, 5F-AB-FUPPYCA remains unscheduled at the federal level. It could fall under the purview of the federal analog act, either due to its pharmacological properties or as a remote structural analog of JWH-307.
However, in North Dakota, a different stance has been taken. On April 27, 2023, North Dakota categorized 5F-AB-FUPPYCA (N-(1-carbamoyl-2-methyl-propyl)-2-(5-fluoropentyl)-5-(4-fluorophenyl)pyrazole-3-carboxamide) as a Schedule I substance. It’s worth noting that this classification does not extend to structural substitutions or other analogs within the pyrazole category, excluding non-fluorinated AB-FUPPYCA analogs, for instance.
FAQ
1. What is 5F-AB-FUPPYCA?
5F-AB-FUPPYCA is a synthetic cannabinoid known for its potential psychoactive properties. It belongs to the group of designer drugs.
2. Is 5F-AB-FUPPYCA regulated in the United States?
As of May 22, 2023, 5F-AB-FUPPYCA is not federally scheduled in the United States. However, it may be subject to regulation under the Federal Analogue Act due to its pharmacological properties or its structural relationship to JWH-307.
3. What is the Federal Analogue Act?
The Federal Analogue Act allows substances chemically similar to controlled substances but not specifically listed to be treated as controlled if they have similar effects or pharmacological properties.
4. How is 5F-AB-FUPPYCA regulated in North Dakota?
In North Dakota, 5F-AB-FUPPYCA was classified as a Schedule I substance on April 27, 2023. This means it is subject to strict state-level control.
5. Does the North Dakota classification include all similar compounds?
No, the North Dakota classification of 5F-AB-FUPPYCA is specific to that compound. It does not encompass structural substitutions or other analogs in the pyrazole category, excluding non-fluorinated AB-FUPPYCA analogs, for example.
6. What are the potential effects or risks associated with 5F-AB-FUPPYCA?
The specific effects and risks of 5F-AB-FUPPYCA may vary and are not extensively documented. It is essential to exercise caution when dealing with synthetic cannabinoids due to potential unknown health risks.
7. Is 5F-AB-FUPPYCA legal for personal use or purchase?
The legal status of 5F-AB-FUPPYCA differs by jurisdiction. It’s crucial to be aware of and adhere to local laws and regulations regarding the possession and use of such substances.
8. Where can I find more information about 5F-AB-FUPPYCA?
For further information, consider consulting official sources, legal documents, or scientific literature. Always prioritize up-to-date and authoritative information when researching synthetic cannabinoids.
References
- Anvisa (July 24, 2023). “Collegiate Board Resolution No. 804 – Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control” [RDC Nº 804 – Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial] (in Brazilian Portuguese). Published in Diário Oficial da União (on July 25, 2023). Archived from the original on August 27, 2023. Accessed on August 27, 2023.
- Girreser U, Rösner P, Vasilev A (July 2016). “A Case Study: Structure Elucidation of the Designer Drug N-(1-amino-3,3-dimethyl-1-oxobutan-2-yl)-1-(5-fluoropentyl)-3-(4-fluorophenyl)-pyrazole-5-carboxamide and the Significance of Predicted (13) C NMR Shifts.” Revealed in Drug Testing and Analysis, Volume 8, Issue 7, Pages 668–675. doi:10.1002/dta.1820. PMID 26012418.
- Banister SD, Longworth M, Kevin R, Sachdev S, Santiago M, Stuart J, et al. (September 2016). “Pharmacological Insights into Valinate and tert-Leucinate Synthetic Cannabinoids: 5F-AMBICA, 5F-AMB, 5F-ADB, AMB-FUBINACA, MDMB-FUBINACA, MDMB-CHMICA, and Their Analogues.” Discovered in ACS Chemical Neuroscience, Volume 7, Issue 9, Pages 1241–1254. doi:10.1021/acschemneuro.6b00137. PMID 27421060.
- “2015. ÉVI EURÓPAI KÁBÍTÓSZER – JELENTÉS – A MONITOROZÁS 20 ÉVE” (PDF). Presented by the Hungarian National Focal Point (NFP) in June 2015. Archived (PDF) from the original on July 24, 2015. Retrieved on July 23, 2015.
- “5-fluoro-3,5-AB-PFUPPYCA.” Information from Cayman Chemical Company. Archived from the original on June 3, 2016. Retrieved on February 11, 2016.
- “Test Details: Result #11908 – Plant Material, 11908.” Available on DrugsData.org (formerly EcstasyData). Archived from the original on April 16, 2022. Retrieved on April 17, 2022.
- “AN ACT to amend and reenact sections 19-03.1-05, 19-03.1-11, and 19-03.1-13 of the North Dakota Century Code, relating to the scheduling of controlled substances; and to declare an emergency” (PDF). Presented by the Sixty-eighth Legislative Assembly of North Dakota in Regular Session on January 3, 2023.