Beautiful Plants For Your Interior
Summary
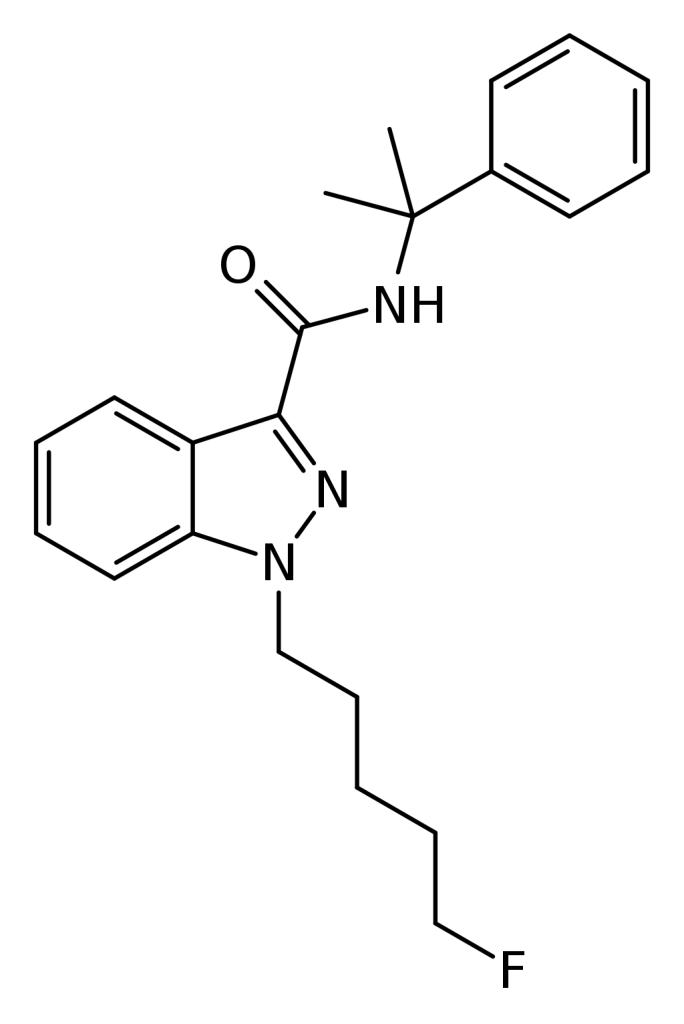
5F-CUMYL-PINACA, also recognized as SGT-25 and occasionally marketed in e-cigarette form as C-Liquid, is a synthetic cannabinoid with an indazole-3-carboxamide base.
This compound acts as a potent agonist for cannabinoid receptors. According to the initial patent, it displayed approximately fourfold selectivity for CB1 receptors, boasting an EC50 of <0.1 nM for human CB1 receptors and 0.37 nM for human CB2 receptors. However, more recent assays using different techniques have yielded varying results. Some studies indicate an EC50 of 0.43 nM at CB1 and 11.3 nM at CB2, suggesting a roughly 26-fold CB1 selectivity. Alternatively, another data set suggests 15.1 nM at CB1 and 34.8 nM at CB2 with only 2.3 times selectivity. It’s worth noting that these figures cannot be directly compared due to the differing assay techniques used in each case.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1400742-16-6 |
|---|---|
| PubChem CID | 86274158 |
| ChemSpider | 35228406 |
| UNII | E827UUV3S9 |
| Chemical and physical data | |
| Formula | C22H26FN3O |
| Molar mass | 367.468 g·mol−1 |
Legal status
5F-CUMYL-PINACA was placed under temporary emergency scheduling by the DEA in the United States in 2019, and subsequently, it was designated as a permanent Schedule I Controlled Substance on April 7, 2022. In Sweden, the national public health agency recommended classifying 5F-CUMYL-PINACA as a hazardous substance on November 10, 2014.
FAQ
1. What is 5F-CUMYL-PINACA?
- 5F-CUMYL-PINACA is a synthetic cannabinoid compound known for its interaction with cannabinoid receptors, producing psychoactive effects similar to those of cannabis.
2. Why did the DEA schedule the 5F-CUMYL-PINACA temporary emergency in the United States?
- The DEA temporarily emergency scheduled 5F-CUMYL-PINACA in 2019 due to concerns about its safety and potential health risks associated with its use.
3. When did 5F-CUMYL-PINACA become a permanent Schedule I Controlled Substance in the United States?
- 5F-CUMYL-PINACA was designated as a permanent Schedule I Controlled Substance on April 7, 2022, by the DEA.
4. What does it mean for a substance to be classified as a Schedule I Controlled Substance in the United States?
- Schedule I substances are classified as the most restrictive category under the Controlled Substances Act. They are considered to have a high potential for abuse and no currently accepted medical use. Possession, distribution, or manufacture of Schedule I substances is illegal.
5. Why did Sweden’s public health agency recommend classifying 5F-CUMYL-PINACA as a hazardous substance?
- Sweden’s public health agency made this recommendation on November 10, 2014, due to concerns about the potential health hazards and risks associated with using 5F-CUMYL-PINACA.
6. Is 5F-CUMYL-PINACA still legal in any country?
- The legal status of 5F-CUMYL-PINACA varies by country and jurisdiction. It is essential to check the specific laws and regulations in your area to determine their legality.
7. What are the potential risks associated with the use of 5F-CUMYL-PINACA?
- The use of 5F-CUMYL-PINACA, like other synthetic cannabinoids, can be associated with various health risks, including addiction, unpredictable side effects, and potential harm to mental and physical health. It’s crucial to exercise caution and be aware of the potential dangers.
8. Where can I find more information and resources about 5F-CUMYL-PINACA?
- For additional information and guidance regarding 5F-CUMYL-PINACA, consider consulting with local health authorities, substance abuse professionals, or drug education resources. Staying informed is essential to make safe and responsible choices.
References
- Angerer V, Franz F, Moosmann B, Bisel P, and Auwärter V conducted a comprehensive study on “5F-Cumyl-PINACA in ‘e-liquids’ for electronic cigarettes.” They characterized this synthetic cannabinoid and investigated its in vitro and in vivo phase I metabolism. Their findings were published in Forensic Toxicology (2018), shedding light on the composition and effects of this trendy product. [Reference: Angerer V, Franz F, Moosmann B, Bisel P, Auwärter V (13 November 2018). “5F-Cumyl-PINACA in ‘e-liquids’ for electronic cigarettes: comprehensive characterization of a new type of synthetic cannabinoid in a trendy product including investigations on the in vitro and in vivo phase I metabolism of 5F-Cumyl-PINACA and its non-fluorinated analog Cumyl-PINACA”. Forensic Toxicology. 37 (1): 186–196. doi:10.1007/s11419-018-0451-8. PMC 6315005. PMID 30636986.]
- “5-fluoro CUMYL-PINACA (CRM)” was mentioned by Cayman Chemical, a prominent entity in the field of chemical research. This reference provides information about this specific synthetic cannabinoid. [Reference: “5-fluoro CUMYL-PINACA (CRM)”. Cayman Chemical. Retrieved 22 November 2018.]
- A New Zealand patent application from Bowden and colleagues (2013) is noted, revealing information on cannabinoid compounds. This source is relevant for understanding the development and chemistry of related substances. [Reference: Bowden; et al. (11 April 2013). “Patent WO 2014167530 – Cannabinoid compounds”. New Zealand Patent application 623626. Retrieved 22 November 2018.]
- Longworth M, Banister SD, Boyd R, and others (2017) delved into the pharmacology of Cumyl-Carboxamide Synthetic Cannabinoids, including CUMYL-5F-PINACA. Their research was published in ACS Chemical Neuroscience, providing insights into the properties of these substances. [Reference: Longworth M, Banister SD, Boyd R, Kevin RC, Connor M, McGregor IS, Kassiou M (October 2017). “Pharmacology of Cumyl-Carboxamide Synthetic Cannabinoid New Psychoactive Substances (NPS) CUMYL-BICA, CUMYL-PICA, CUMYL-5F-PICA, CUMYL-5F-PINACA, and Their Analogues”. ACS Chemical Neuroscience. 8 (10): 2159–2167. doi:10.1021/acschemneuro.7b00267. PMID 28792725.]
- Asada A, Doi T, Tagami T, and colleagues (2018) explored the cannabimimetic activities of cumyl carboxamide-type synthetic cannabinoids. This research, published in Forensic Toxicology, contributes to understanding the effects of these substances. [Reference: Asada A, Doi T, Tagami T, Takeda A, Satsuki Y, Kawaguchi M, Nakamura A, Sawabe Y (January 2018). “Cannabimimetic activities of cumyl carboxamide-type synthetic cannabinoids”. Forensic Toxicology. 36 (1): 170–177. doi:10.1007/s11419-017-0374-9. ISSN 1860-8965. S2CID 7996915.]
- In a Federal Register notice from April 2019, it was stated that “5F-CUMYL-PINACA” was temporarily placed in Schedule I under controlled substances. This designation is part of regulatory efforts in the United States. [Reference: “Schedules of Controlled Substances: Temporary Placement of 5F-EDMB-PINACA, 5F-MDMB-PICA, FUB-AKB48, 5F-CUMYL-PINACA, and FUB-144 into Schedule I”. Federal Register. 16 April 2019.]
- Another Federal Register notice from April 2022 mentioned the placement of “5F-CUMYL-PINACA” in Schedule I as a controlled substance, making its status permanent in the United States. [Reference: “Schedules of Controlled Substances: Placement of 5F-EDMB-PINACA, 5FMDMB-PICA, FUB-AKB48, 5F-CUMYLPINACA, and FUB-144 in Schedule I” (PDF). Federal Register. 87 (67). 7 April 2022.]
- Sweden’s Public Health Agency recommended classifying cannabinoids as health-endangering substances in 2014, reflecting concerns about their potential risks. [Reference: “Cannabinoider föreslås bli klassade som hälsofarlig vara” [Cannabinoids suggested classified as health endangering substance] (in Swedish). Public Health Agency of Sweden. 10 November 2014. Retrieved 11 July 2015.]