Beautiful Plants For Your Interior
Summary
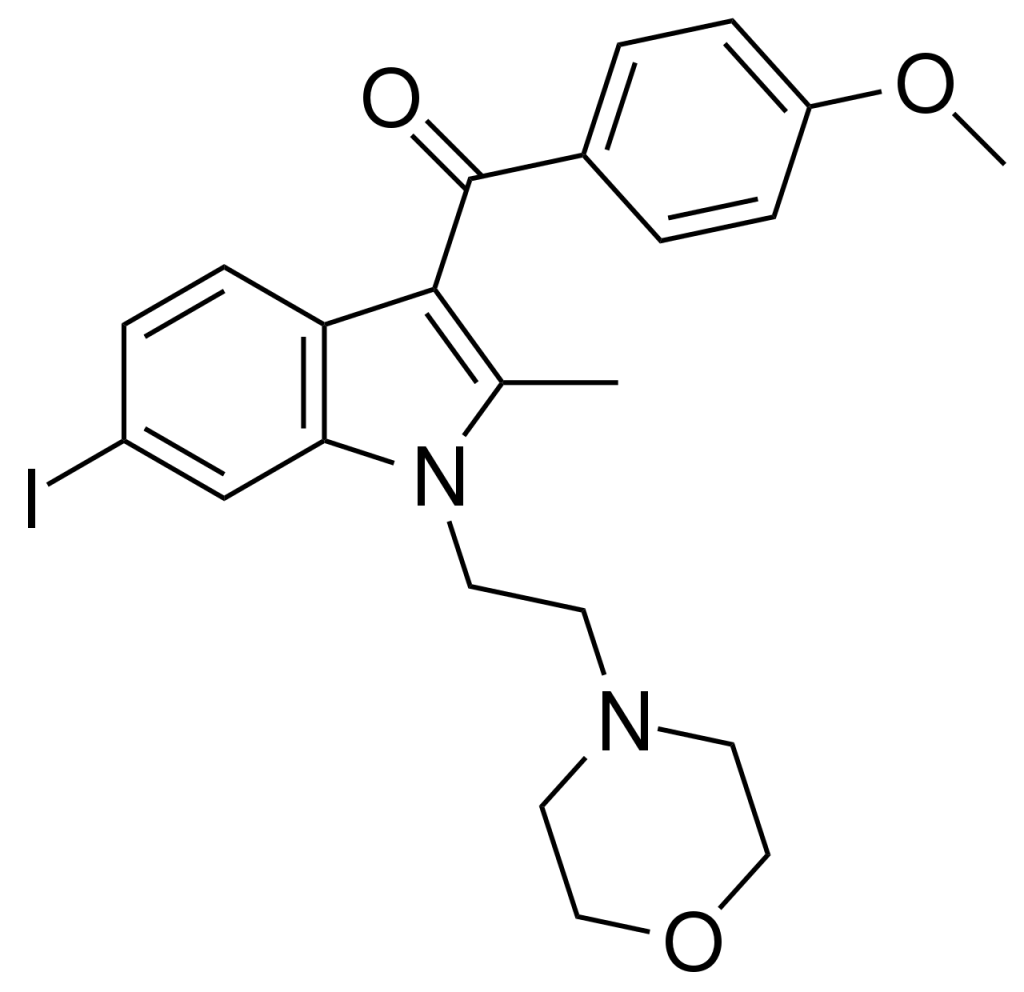
AM-630, or 6-Iodopravadoline, is a potent and highly selective inverse agonist targeting the cannabinoid receptor CB2. It exhibits a remarkable affinity for CB2, boasting a Ki value of 32.1 nM, while showing a notable 165-fold selectivity over CB1 receptors, where it only exhibits weak partial agonist activity. Researchers employ AM-630 to investigate CB2-mediated responses, shedding light on the potential involvement of CB2 receptors in brain function. Notably, AM-630 holds significance as one of the pioneering indole-derived cannabinoid ligands that feature substitution at the 6-position of the indole ring. This particular substitution has since been recognized as a crucial determinant for affinity and efficacy at both CB1 and CB2 receptors, consequently paving the way for the development of numerous related derivatives.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 164178-33-0 |
|---|---|
| PubChem CID | 4302963 |
| IUPHAR/BPS | 750 |
| ChemSpider | 3508738 |
| UNII | U1LNJ6NBKA |
| ChEMBL | ChEMBL181633 |
| CompTox Dashboard (EPA) | DTXSID10167719 |
| ECHA InfoCard | 100.229.964 |
| Chemical and physical data | |
| Formula | C23H25IN2O3 |
| Molar mass | 504.368 g·mol−1 |
FAQ
- What is AM-630 (6-Iodopravadoline)?
- AM-630 is a chemical compound that acts as a potent and selective inverse agonist for the cannabinoid receptor CB2.
- What is the role of AM-630 in the endocannabinoid system?
- AM-630 interacts with the CB2 receptor, a part of the endocannabinoid system. It exhibits inverse agonist properties, meaning it reduces the activity of the CB2 receptor, as opposed to an agonist, which would enhance its activity.
- What is the Ki value of AM-630 at the CB2 receptor?
- AM-630 has a Ki (binding affinity) of 32.1 nM at the CB2 receptor. This value reflects its high affinity for this receptor.
- How selective is AM-630 for CB2 over CB1?
- AM-630 is highly selective, with a selectivity of 165 times for the CB2 receptor over the CB1 receptor. This selectivity is one of its notable features.
- What is the significance of AM-630 in research?
- AM-630 is used in the study of CB2-mediated responses and is valuable for investigating the potential role of CB2 receptors in the brain. It helps researchers better understand the functions of the endocannabinoid system.
- What structural features of AM-630 make it essential in cannabinoid research?
- AM-630 is one of the first indole-derived cannabinoid ligands with a substitution at the 6-position of the indole ring. This structural feature has since been recognized as crucial for determining affinity and efficacy at CB1 and CB2 receptors, leading to the development of related derivatives.
- How can AM-630 be obtained for research purposes?
- Researchers typically obtain AM-630 through specialized chemical suppliers or by collaborating with research institutions with the necessary permits to acquire controlled substances.
- Are there any known therapeutic applications for AM-630?
- As of my last knowledge update in January 2022, AM-630 is primarily used for research and has no approved therapeutic applications. The field of study and drug development constantly evolves, so you may want to check for any updates on potential medicinal uses.
- Is AM-630 legal to possess or use?
- The legality of AM-630 may vary by jurisdiction and its intended use. It’s essential to ensure compliance with local, national, and international regulations when working with substances like AM-630, mainly because it may be classified as a controlled substance in some areas.
- Where can I find more information about AM-630?
- You can find additional information about AM-630 and related research in scientific journals, academic databases and by consulting experts in cannabinoid pharmacology and research. Always follow appropriate safety and legal guidelines when researching or working with such substances.
References
- In a study by Ross RA and colleagues (February 1999), the compounds L759633, L759656, and AM630 were characterized as agonists and inverse agonists at CB1 and CB2 cannabinoid receptors. Their findings were published in the “British Journal of Pharmacology” (126(3), 665–672), shedding light on the pharmacological properties of these compounds.
- Murataeva N, Mackie K, and Straiker A (November 2012) investigated the CB2-preferring agonist JWH015 and its effects on CB1 activation in autaptic hippocampal neurons. Their research, published in “Pharmacological Research” (66(5), 437–442), uncovered significant insights into the interplay between CB1 and CB2 receptors in neuronal systems.
- Morgan NH, Stanford IM, and Woodhall GL (September 2009) explored the presence of functional CB2 type cannabinoid receptors at central nervous system (CNS) synapses. Their study, featured in “Neuropharmacology” (57(4), 356–368), contributed to our understanding of cannabinoid receptor functionality within the CNS.
- Ishiguro H and collaborators (May 2010) delved into the role of the brain cannabinoid CB2 receptor in schizophrenia. Their work, published in “Biological Psychiatry” (67(10), 974–982), examined the potential implications of CB2 receptors in the context of this mental disorder.
- Eissenstat MA and colleagues (August 1995) investigated the structure-activity relationships of novel cannabinoid mimetics known as aminoalkylindoles. Their research, documented in the “Journal of Medicinal Chemistry” (38(16), 3094–3105), provided valuable insights into the development of cannabinoid-related compounds.
- Hongfeng Deng’s PhD dissertation (2000) at the University of Connecticut focused on the design and synthesis of selective cannabinoid receptor ligands, particularly aminoalkylindole and other heterocyclic analogs. This academic work expanded our knowledge of cannabinoid receptor ligand development.
- Hynes J and collaborators (September 2002) delved into the modulation of cannabinoid receptors using C-3 Amido-indole compounds. Their research, published in “Bioorganic & Medicinal Chemistry Letters” (12(17), 2399–2402), explored the potential of these compounds as modulators of cannabinoid receptors.
- Frost JM and colleagues (March 2008) investigated the effects of indole ring substitution on CB2 cannabinoid receptor activity using indol-3-yl-tetramethylcyclopropyl ketones. Their findings, featured in the “Journal of Medicinal Chemistry” (51(6), 1904–1912), contributed to our understanding of the structural determinants of CB2 receptor activity.
- In 2010, Adam JM and his team focused on the design, synthesis, and structure-activity relationships of indole-3-carboxamides as novel water-soluble cannabinoid CB1 receptor agonists. Their research, published in “MedChemComm” (1, 54), explored the development of water-soluble compounds targeting CB1 receptors, offering potential therapeutic implications.