4F-MPH, a synthetic stimulant and designer drug, has garnered attention among researchers and enthusiasts alike. However, the ease of its availability online from various research chemical sellers raises significant concerns regarding safety, quality control, and ethical considerations.
The online market for research chemicals has exploded in recent years, allowing individuals to buy substances like 4F-MPH with relative ease. While some vendors may claim to provide high-quality products for research purposes, the reality often needs to be improved. One of the major issues with buying 4F-MPH online is the need for more regulation and oversight, which means that the purity and safety of the substance can vary widely from one seller to another.
One of the critical problems with these online 4F-MPH vendors is the potential for mislabeling or adulteration of the product. Researchers and users are at risk of receiving a different chemical altogether, which can have serious health consequences. Moreover, the inconsistency in product quality from other sellers makes it difficult to conduct reliable scientific experiments or draw meaningful conclusions from research.
Another issue is the ethical dilemma surrounding the sale of 4F-MPH and other research chemicals online. These substances are often marketed as “not for human consumption,” but the reality is that they are often purchased and used for recreational purposes. This blurs the line between legitimate research and recreational drug use, which can have legal and societal implications.
Contents
- 1 Summary
- 2 Chemistry
- 3 Pharmacology
- 4 Subjective effects
- 5 Toxicity
- 6 Legal status
- 7 FAQ
- 7.1 1. What is 4F-MPH?
- 7.2 2. What are the common street names for 4F-MPH?
- 7.3 3. Is 4F-MPH legal?
- 7.4 4. How is 4F-MPH typically used?
- 7.5 5. What are the effects of 4F-MPH?
- 7.6 6. Are there any health risks associated with 4F-MPH use?
- 7.7 7. Is 4F-MPH addictive?
- 7.8 8. What precautions should I take if I choose to use 4F-MPH?
- 7.9 9. Can 4F-MPH be detected in drug tests?
- 8 References
Summary
4-Fluoromethylphenidate, commonly referred to as 4F-MPH, belongs to the phenidate class of synthetic stimulants. When administered, it produces prolonged feelings of euphoria and stimulation. This compound closely resembles methylphenidate, a well-known medication used to treat ADHD under brand names such as Ritalin and Concerta.
Both 4F-MPH and methylphenidate are believed to operate through similar pharmacological mechanisms as monoamine reuptake inhibitors. However, they are reported to elicit distinct subjective effects. Notably, 4F-MPH is often described as significantly more euphoric and recreational. Anecdotal accounts indicate it is more potent and tends to cause fewer uncomfortable side effects like anxiety, muscle spasms, and the urge to re-dose. This heightened potency could be attributed to its demonstrated capacity to function as a more efficient dopamine reuptake inhibitor than its parent compound, methylphenidate.
4F-MPH has a relatively short history of recreational use and has yet to be documented as being sold on the streets. Initially, it was developed as a substitute for methylphenidate, which was prohibited in the United Kingdom in April 2015 following a temporary blanket ban. Subsequently, it became available for purchase on the online grey market, where it is marketed as a research chemical intended for global distribution.
Given its potent and long-lasting stimulant effects, the potential for habit-forming properties and an unknown toxicity profile, it is strongly advised that individuals exercise caution and adopt proper harm-reduction practices if they choose to use this substance.
| Identifiers | |
|---|---|
| show IUPAC name | |
| CAS Number | 1354631-33-6 |
| PubChem CID | 70876096 |
| ChemSpider | 26350585 |
| UNII | TN2LV2C0X9 |
| Chemical and physical data | |
| Formula | C14H18FNO2 |
| Molar mass | 251.301 g·mol−1 |
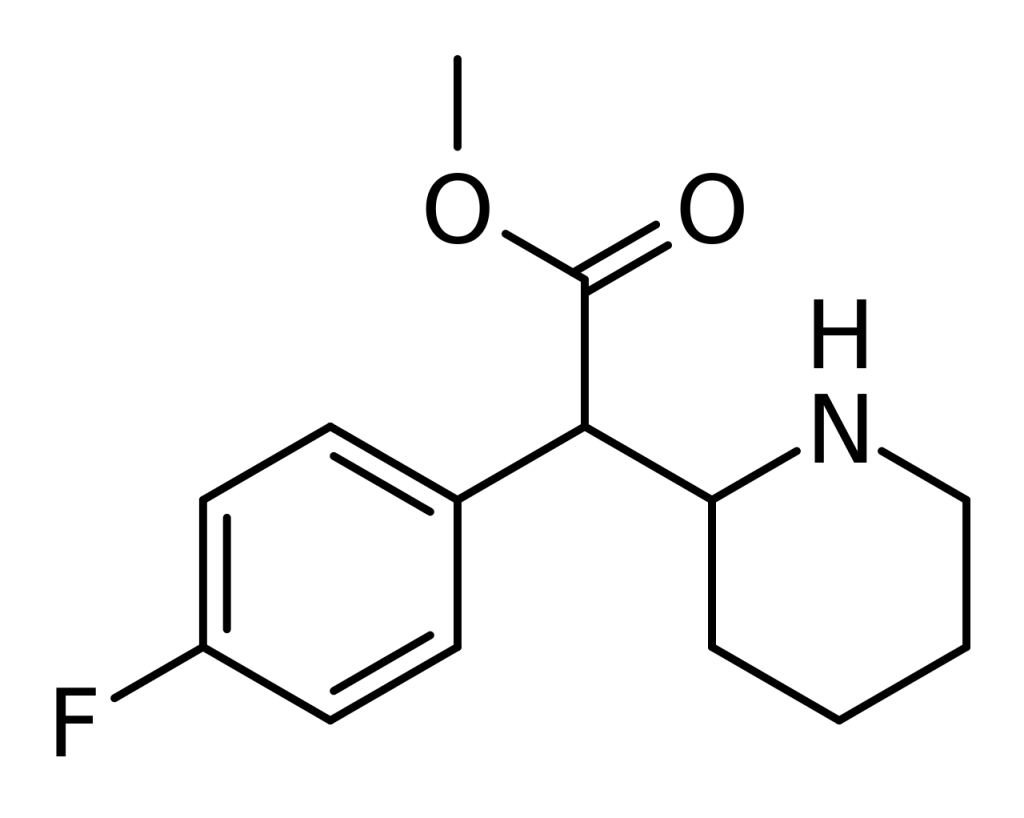
Chemistry
4F-MPH is a synthetic compound belonging to the substituted phenethylamine and phenidate categories. It is a fluorinated derivative of methylphenidate, characterized by a phenethylamine core. This core comprises a phenyl ring connected to an amino (-NH2) group through an ethyl chain. Structurally, it shares similarities with amphetamine, featuring a substitution at the Rα position that forms a piperidine ring, culminating at the terminal amine of the phenethylamine chain. Additionally, it incorporates a methyl acetate group bound to the Rβ position within its structure. The primary distinction between 4F-MPH and methylphenidate lies in the presence of a single fluorine atom attached to the four-position of the phenethylamine core.
Regarding terminology, the “methyl-” in methylphenidate pertains to the side chain composed of a single carbon atom, while “Phen-” signifies the phenyl ring. The term “id-” is derived from a piperidine ring, and “-ate” denotes the presence of the acetate group containing oxygen atoms. Like its parent molecule, 4F-MPH is a chiral compound typically produced as a racemic mixture.
A noteworthy scientific observation is that amphetamine analogues incorporating fluorine, chlorine, bromine, and methyl groups tend to exhibit greater potency compared to those lacking these substitutions. This structural insight underscores the potential impact of fluorination on the pharmacological properties of such compounds.
Pharmacology
4F-MPH is believed to function as an inhibitor of dopamine and norepinephrine reuptake primarily. This means that it effectively enhances the levels of these neurotransmitters in the brain by binding to and partially obstructing the transporter proteins responsible for clearing dopamine and norepinephrine from the synaptic cleft. Consequently, this accumulation of dopamine and norepinephrine within the brain’s reward pathways leads to stimulating and euphoric effects.
In a pharmacological assessment, the (±)-three isomer of 4F-MPH was found to be approximately 2.15 times more potent in inhibiting dopamine reuptake and 2.7 times more powerful in inhibiting norepinephrine reuptake compared to its parent compound, methylphenidate. Conversely, the (±)-erythro isomer exhibited significantly lower effectiveness, being approximately 65 times less potent in dopamine reuptake inhibition and about 45.6 times less potent in norepinephrine reuptake inhibition compared to methylphenidate. Notably, neither the racemate of 4F-MPH significantly influenced serotonin reuptake.
Subjective effects
Disclaimer: The ensuing effects, as detailed below, are based on the Subjective Effect Index (SEI), a compilation of anecdotal user reports, and the personal observations of PsychonautWiki contributors. Consequently, readers are urged to approach these descriptions with a discerning perspective.
Acknowledging that these effects may not necessarily manifest consistently or predictably is essential. However, it is noteworthy that higher doses are more likely to yield the complete spectrum of products. Conversely, adverse effects become increasingly probable with higher doses and may encompass addiction, severe harm, or even fatality ☠.
Physical:
- Stimulation: 4F-MPH is typically characterized as energetic and stimulating. Its stimulant properties are discernible but milder than amphetamine or methamphetamine and stronger than methylphenidate, modafinil, and caffeine. Lower to moderate doses promote productivity, while higher doses can induce physical activities such as dancing, socializing, running, or cleaning. The style of stimulation is often described as “forced,” leading to jaw clenching, involuntary bodily shakes, and vibrations, resulting in extreme body trembling, unsteady hands, and reduced motor control. This stimulation closely resembles that of methylphenidate.
- Dehydration
- Nausea: This may occur at higher doses but typically subsides relatively quickly.
- Appetite suppression: Less intense than amphetamine and methylphenidate.
- Increased heart rate
- Abnormal heartbeat
- Increased perspiration
- Teeth grinding: Comparatively less intense than MDMA.
- Vasoconstriction
- Increased libido: Higher doses of 4F-MPH may enhance sexual desire, though this effect is considered less intense and consistent than amphetamines and cocaine but more potent than regular methylphenidate.
- Dizziness may occur at higher doses but tends to diminish after a brief period.
- Spontaneous physical sensations
- Pupil dilation
After:
- Anxiety
- Cognitive fatigue: This can manifest as a rebound effect during the offset of the compound, often matching the intensity of the enhancements experienced earlier.
- Depression
- Irritability
- Motivation suppression
- Thought deceleration
- Wakefulness
Cognitive:
- Anxiety: Reported as more robust and frequent than with other commonly used stimulants like amphetamine or cocaine, similar to the closely related compound methylphenidate.
- Analysis enhancement
- Euphoria: The euphoric rush linked to 4F-MPH use (attributable to dopamine reuptake inhibition) is fleeting and compulsive, akin to cocaine, but typically less consistent and intense.
- Compulsive redosing: Occurs with less frequency than other common stimulants like amphetamine or cocaine but becomes more prevalent at high doses or non-oral administration. Setting a limit or opting for oral dosing is advised.
- Ego inflation
- Emotion suppression: More pronounced than other stimulants, aligning with observed emotion-suppressing effects in stimulants, especially methylphenidate and related compounds.
- Focus enhancement: Most effective at low to moderate dosages, as higher doses tend to impair concentration.
- Increased music appreciation
- Time distortion: Involves the perception of time passing much faster than usual when sober.
- Motivation enhancement
- Memory enhancement
- Suggestibility suppression
- Thought acceleration
- Thought organization: Often more noticeable than other stimulants, prompting some users to employ it for productivity or functional purposes rather than recreational ones.
- Wakefulness
Toxicity
The scientific exploration of the toxicity and potential long-term health consequences associated with recreational 4F-MPH usage has yet to be undertaken, as this research chemical lacks a substantial history of human consumption. Consequently, the precise toxic dosage remains unknown. Anecdotal feedback from individuals who have experimented with 4F-MPH suggests no apparent adverse health effects are linked to cautious, low to moderate dosage usage. However, it’s essential to remember that no guarantees can be provided. Always conduct independent research to ensure the safety of combining two or more substances before consumption.
The utmost importance is placed on using harm-reduction practices when employing this substance.
Tolerance and Potential for Addiction:
4F-MPH can be used consecutively for extended durations, but it is subject to acute tolerance, which gradually escalates with prolonged usage. Consequently, users may need to increase their dosage to attain the same effects. It’s worth noting that tolerance tends to develop more rapidly in comparison to methylphenidate.
Due to its minimal tolerance development, euphoric effects, and impact on dopamine transporters, 4F-MPH has an abuse potential on par with amphetamine or MDMA.
Dangerous Interactions:
A word of caution: several psychoactive substances, individually relatively safe, can suddenly become hazardous, even life-threatening, when combined with specific other substances. The ensuing list outlines some known perilous interactions, though it may not encompass all possibilities.
Always engage in independent research (e.g., through Google, DuckDuckGo, PubMed) to verify the safety of consuming a combination of two or more substances. Some of the interactions listed are derived from TripSit.
- 25x-NBOMe & 25x-NBOH: These compounds are highly stimulating and physically taxing. Combining them with 4F-MPH should be avoided due to the risk of excessive stimulation, heart strain, heightened blood pressure, vasoconstriction, panic attacks, thought loops, seizures, and, in extreme cases, heart failure.
- Alcohol: The combination of alcohol with stimulants poses dangers due to the potential for unintentional over-intoxication. Stimulants mask the depressant effects of alcohol, making it challenging for individuals to assess their level of intoxication. Once the stimulant’s effects diminish, the depressant effects can emerge unchecked, potentially leading to blackouts and severe respiratory depression. If alcohol and stimulants are combined, users should strictly limit their alcohol consumption per hour.
- DXM: Combining DXM with 4F-MPH should be avoided due to DXM’s inhibitory effects on serotonin and norepinephrine reuptake. This combination heightens the risk of panic attacks, hypertensive crisis, or serotonin syndrome when used with serotonin releasers like MDMA, methylone, or mephedrone. Monitoring blood pressure carefully and abstaining from strenuous physical activity is essential.
- MDMA: Other stimulants can amplify any potential neurotoxic effects associated with MDMA. Additionally, there is a risk of elevated blood pressure and heart strain (cardiotoxicity).
- MXE: Reports suggest combining MXE with 4F-MPH can dangerously elevate blood pressure and increase the likelihood of mania and psychosis.
- Dissociatives: Both 4F-MPH and dissociatives carry the risk of inducing delusions, mania, and psychosis, which may be compounded when used together.
- Stimulants: Combining 4F-MPH with other motivations, such as cocaine, can lead to dangerously elevated heart rates and blood pressure levels.
- Tramadol: Tramadol is known to lower the seizure threshold, and combining it with stimulants may further heighten this risk.
- MAOIs: This combination can elevate neurotransmitter levels, such as dopamine, to dangerous or fatal levels. Examples of MAOIs include Syrian rue, banisteriopsis caapi, and certain antidepressants.
Legal status
Germany: 4-Fluoromethylphenidate was classified as a controlled substance under the NpSG (New Psychoactive Substances Act) as of November 26, 2016. However, it unintentionally regained legal status in September 2022 and is currently considered legal again.
Switzerland: 4F-MPH is categorized as a controlled substance, listed explicitly in Verzeichnis E.
Turkey: 4F-MPH is classified as a drug and is illegal to possess, produce, supply, or import.
United Kingdom: 4-Fluoromethylphenidate is classified as a class B drug in the UK as of May 31, 2017, and is illegal to possess, produce, or supply.
United States: 4-Fluromethylphenidate is designated as a Schedule I controlled substance in Alabama.
FAQ
1. What is 4F-MPH?
4F-MPH, short for 4-Fluoromethylphenidate, is a synthetic stimulant belonging to the phenidate class. It is structurally similar to methylphenidate, commonly used to treat ADHD (Attention Deficit Hyperactivity Disorder).
2. What are the common street names for 4F-MPH?
Street names for 4F-MPH may include “4F,” “4F Ritalin,” or simply “Methylphenidate.”
3. Is 4F-MPH legal?
The legality of 4F-MPH varies by country and region. You must check local laws and regulations regarding its possession, sale, or use.
4. How is 4F-MPH typically used?
4F-MPH is usually taken orally, but some individuals may choose to insufflate (snort) it. It is often used recreationally or as a study aid due to its stimulating effects.
5. What are the effects of 4F-MPH?
4F-MPH produces stimulating effects, including increased energy, alertness, and enhanced focus. It may also induce euphoria, although this effect tends to be short-lived.
6. Are there any health risks associated with 4F-MPH use?
The long-term health effects of 4F-MPH are not well-studied, and its safety profile is not fully understood. Like other stimulants, it can lead to potential risks such as increased heart rate, high blood pressure, anxiety, and addiction if used excessively.
7. Is 4F-MPH addictive?
4F-MPH has the potential for abuse and addiction, mainly when used regularly or at high doses. Users should exercise caution and be aware of the risk of developing dependence.
8. What precautions should I take if I choose to use 4F-MPH?
If you decide to use 4F-MPH, it is essential to follow harm reduction practices. Start with a low dose, avoid mixing it with other substances, stay hydrated, and be mindful of potential side effects.
9. Can 4F-MPH be detected in drug tests?
4F-MPH can appear on drug tests, particularly those that screen for amphetamines or stimulants. If you are subject to drug testing, be aware of the substances that may be detected.
References
- In a study conducted by Deutsch, H. M. and colleagues in March 1996, they delved into the “Synthesis and Pharmacology of Potential Cocaine Antagonists.” Their research, published in the Journal of Medicinal Chemistry, explored the structure-activity relationship studies of aromatic ring-substituted methylphenidate analogues, shedding light on potential cocaine antagonists and their properties.
- Schwerin, M. M., Deutsch, H. M., Massey, A. T., and Holtzman, S. G., in a May 2002 publication in The Journal of Pharmacology and Experimental Therapeutics, presented “Biochemical and behavioural characterization of novel methylphenidate analogues.” This work provided valuable insights into the biochemical and behavioural aspects of new methylphenidate analogues.
- Davies, H. M. L., Hopper, D. W., Hansen, T., Liu, Q., and Childers, S. R., on April 5, 2004, contributed to the field with their research on the “Synthesis of methylphenidate analogues and their binding affinities at dopamine and serotonin transport sites.” This study, published in Bioorganic & Medicinal Chemistry Letters, highlighted the binding affinities of methylphenidate analogues at critical transport sites.
- Misra, M., Shi, Q., Ye, X., Gruszecka-Kowalik, E., Bu, W., Liu, Z., Schwerin, M. M., Deutsch, H. M., and Venanzi, C. A., in October 2010, conducted “Quantitative structure-activity relationship studies of three-methylphenidate analogues.” Their research, featured in Bioorganic & Medicinal Chemistry, focused on the quantitative relationships between the structure of the methylphenidate analogues and their activities.
- In a March 2000 publication titled “Chemistry, Design, and Structure−Activity Relationship of Cocaine Antagonists,” Singh provided an in-depth analysis of cocaine antagonists, their design, and the structure-activity relationships that govern their functionality. This work was published in Chemical Reviews.
- McLaughlin, G., Morris, N., Kavanagh, P. V., Power, J. D., Dowling, G., Twamley, B., O’Brien, J., Hessman, G., Murphy, B., Walther, D., Partilla, J. S., Baumann, M. H., and Brandt, S. D., in March 2017, conducted analytical characterization and pharmacological evaluation of the new psychoactive substance 4-fluoromethylphenidate (4F-MPH). Their work, published in Drug Testing and Analysis, also involved distinguishing between the (±)- three and (±)- erythro diastereomers of 4F-MPH.
- Talaie, H., Panahandeh, R., Fayaznouri, M. R., Asadi, Z., and Abdollahi, M., in 2009, investigated the occurrence of seizures with tramadol. Their research, published in the Journal of Medical Toxicology, revealed the dose-independent occurrence of seizures associated with tramadol use.
- Gillman, P. K. 2005 explored the interaction between monoamine oxidase inhibitors, opioid analgesics, and serotonin toxicity in a publication titled “Monoamine oxidase inhibitors, opioid analgesics and serotonin toxicity” in the British Journal of Anaesthesia. This work discussed the potential risks associated with the combination of these substances.
- “Anlage NpSG,” a German document, can be found on the Bundesministerium der Justiz und für Verbraucherschutz website, and it relates to the control of new psychoactive substances [9].
- The “Gesetz zur Bekämpfung der Verbreitung neuer psychoaktiver Stoffe” is a German law aimed at combating the spread of new psychoactive substances, available in PDF format from the Bundesanzeiger Verlag.
- The “Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien” is a Swiss regulation governing the lists of controlled substances, accessible from the Bundeskanzlei [11].
- For official information on Turkish legislation, “Cumhurbaşkanı Kararı Karar Sayısı: 1335” can be referenced from the official government gazette, resmigazete.gov.tr.
- The “Misuse of Drugs Act 1971 (Amendment) Order 2017” is a legal document related to drug regulation in the UK.
- “Alabama SB333, 2014, Regular Session” is a legislative document from Alabama, USA, related to drug policy.