Beautiful Plants For Your Interior
Summary
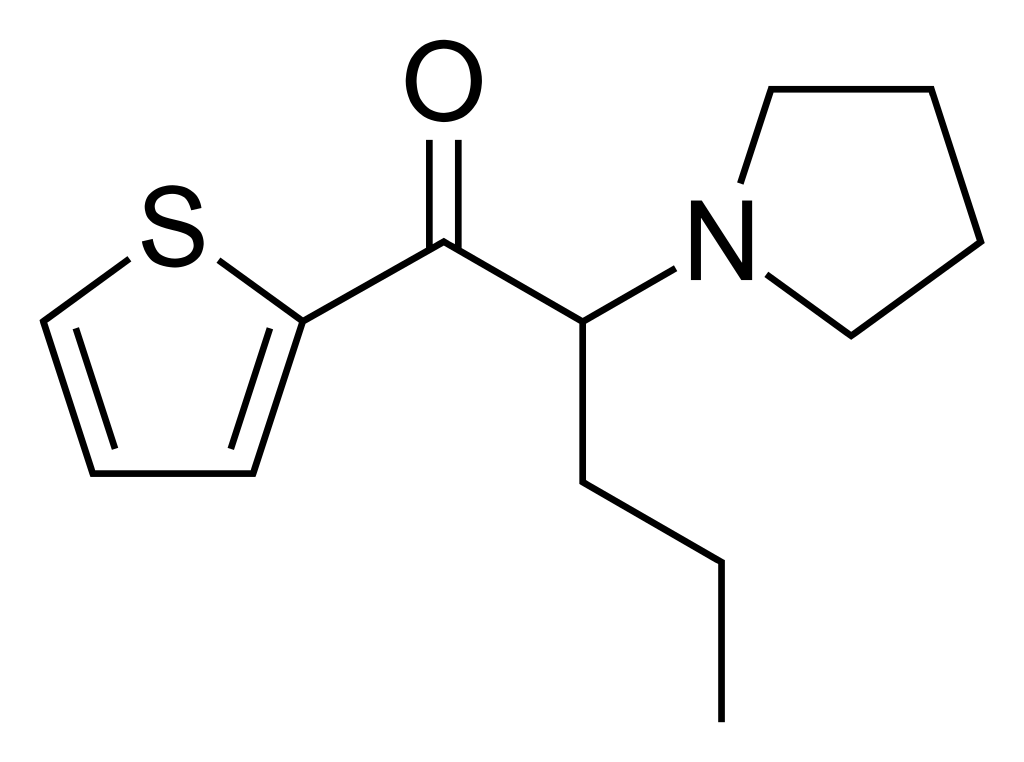
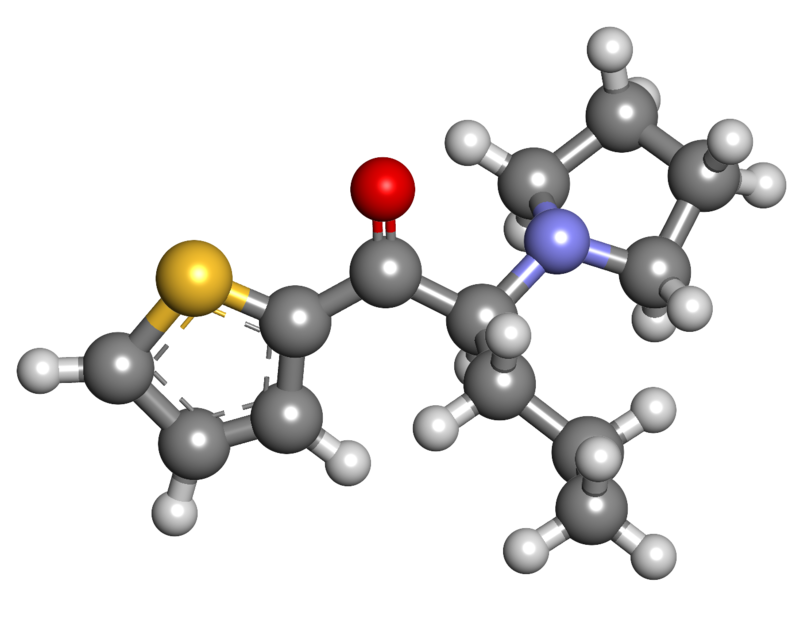
α-Pyrrolidinopentiophenone (α-PVT), also known as α-PVT, is a synthetic stimulant in the cathinone class. This compound has been available for purchase as a designer drug on online platforms. It is structurally related to α-PVP, with the key distinction being the replacement of the phenyl ring with a thiophene ring.
The initial discovery of α-PVT occurred in Japan in 2013. Since then, its metabolic pathways and characteristics have been documented in scientific literature.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1400742-66-6 |
|---|---|
| PubChem CID | 71721462 |
| ChemSpider | 29738196 |
| UNII | 6JA2K09914 |
| CompTox Dashboard (EPA) | DTXSID90857020 |
| Chemical and physical data | |
| Formula | C13H19NOS |
| Molar mass | 237.36 g·mol−1 |
Side effects
Research has demonstrated that α-PVT exhibits significant cytotoxicity when tested against human cell lines.
Legality
On November 10, 2014, Sweden’s public health agency recommended categorizing α-PVT as a hazardous substance. Furthermore, starting from October 2015, α-PVT has been classified as a controlled substance in China. Switzerland also prohibited the possession and use of α-PVT as of December 2015.
FAQ
1. What is α-Pyrrolidinopentiophenone (α-PVT)?
α-Pyrrolidinopentiophenone, often referred to as α-PVT, is a synthetic stimulant belonging to the cathinone class. It has been marketed and sold as a designer drug in various regions.
2. What distinguishes α-PVT from other substances in the cathinone class?
α-PVT is structurally similar to α-PVP but features a thiophene ring in place of the phenyl ring, setting it apart from other cathinones.
3. When was α-PVT first discovered?
α-PVT was initially identified in Japan in the year 2013.
4. What is known about the cytotoxicity of α-PVT?
Scientific research has revealed that α-PVT possesses high cytotoxicity when tested against human cell lines.
5. How is α-PVT regulated in different countries?
- In Sweden, the public health agency recommended classifying α-PVT as a hazardous substance on November 10, 2014.
- As of October 2015, China has classified α-PVT as a controlled substance.
- In Switzerland, α-PVT has been illegal since December 2015.
Please note that the legal status and regulations regarding α-PVT may vary by country, and it is essential to stay informed about local laws and restrictions.
References
- “α-Pyrrolidinopentiothiophenone”. Cayman Chemical. Retrieved on June 29, 2015.
- Concheiro M, Castaneto M, Kronstrand R, Huestis MA (June 2015). “Simultaneous determination of 40 novel psychoactive stimulants in urine by liquid chromatography-high resolution mass spectrometry and library matching”. Journal of Chromatography A. 1397 (5): 32–42. doi:10.1016/j.chroma.2015.04.002. PMC 4433760. PMID 25931378.
- Takahiro Doi; Akiko Asada; Akihiro Takeda; Takaomi Tagami; Munehiro Katagi; Shuntaro Matsuta; Hiroe Kamata; Masami Kawaguchi; Yuka Satsuki; Yoshiyuki Sawabe; Hirotaka Obana (January 2016). “Identification and characterization of α-PVT, α-PBT, and their bromothienyl analogs found in illicit drug products”. Forensic Toxicology. 34 (1): 76–93. doi:10.1007/s11419-015-0288-3. S2CID 24528511.
- Nahoko Uchiyama; Satoru Matsuda; Maiko Kawamura; Ruri Kikura-Hanajiri; Yukihiro Goda (July 2013). “Two new-type cannabimimetic quinolinyl carboxylates, QUPIC and QUCHIC, two new cannabimimetic carboxamide derivatives, ADB-FUBINACA and ADBICA, and five synthetic cannabinoids detected with a thiophene derivative α-PVT and an opioid receptor agonist AH-7921 identified in illegal products”. Forensic Toxicology. 31 (2): 223–240. doi:10.1007/s11419-013-0182-9. S2CID 1279637.
- Takayama T, Suzuki M, Todoroki K, Inoue K, Min JZ, Kikura-Hanajiri R, et al. (June 2014). “UPLC/ESI-MS/TS-based determination of metabolism of several new illicit drugs, ADB-FUBINACA, AB-FUBINACA, AB-PINACA, QUPIC, 5F-QUPIC, and α-PVT, by human liver microsome”. Biomedical Chromatography. 28 (6): 831–8. doi:10.1002/bmc.3155. PMID 24861751.
- Swortwood MJ, Carlier J, Ellefsen KN, Wohlfarth A, Diao X, Concheiro-Guisan M, et al. (January 2016). “In vitro, in vivo and in silico metabolic profiling of α-pyrrolidinopentiothiophenone, a novel thiophene stimulant”. Bioanalysis. 8 (1): 65–82. doi:10.4155/bio.15.237. PMC 5493968. PMID 26648097.
- Wojcieszak J, Andrzejczak D, Woldan-Tambor A, Zawilska JB (August 2016). “Cytotoxic Activity of Pyrovalerone Derivatives, an Emerging Group of Psychostimulant Designer Cathinones”. Neurotoxicity Research. 30 (2): 239–50. doi:10.1007/s12640-016-9640-6. PMID 27295059. S2CID 11202107.
- “Cannabinoider föreslås bli klassade som hälsofarlig vara” (in Swedish). Retrieved on June 29, 2015.
- “关于印发《非药用类麻醉药品和精神药品列管办法》的通知” (in Chinese). China Food and Drug Administration. September 27, 2015. Archived from the original on October 1, 2015. Retrieved on October 1, 2015.
- “Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien” (in German). Der Bundesrat.