Summary
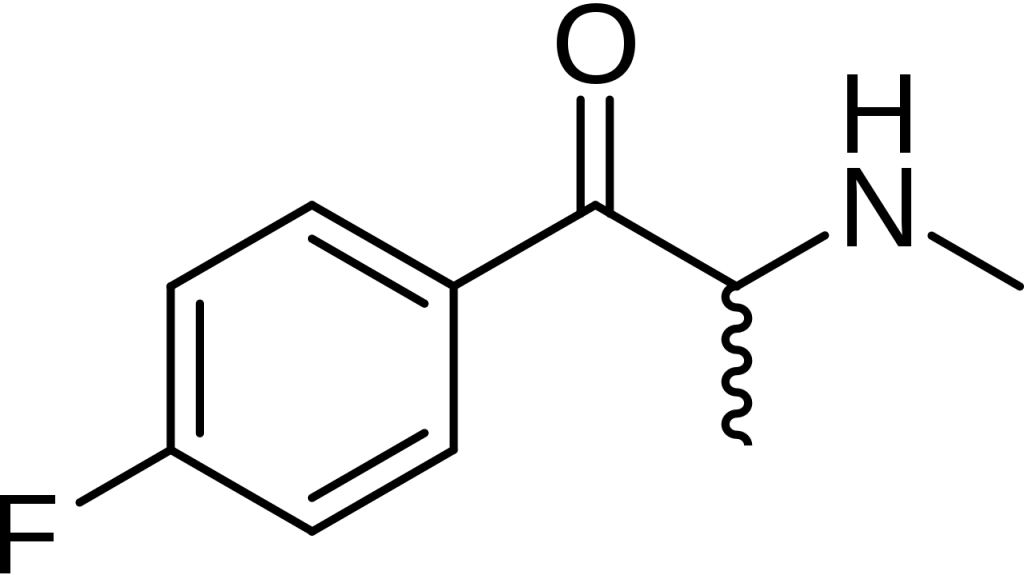
Flephedrone, scientifically called 4-fluoro methcathinone (4-FMC), is a stimulant belonging to the cathinone chemical class. It gained popularity as a designer drug and became available for online purchase, with its emergence dating back to the year 2008.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 447-40-5 |
|---|---|
| PubChem CID | 49853406 |
| ChemSpider | 21477355 |
| UNII | BA88IEZ2HF |
| CompTox Dashboard (EPA) | DTXSID401028417 |
| Chemical and physical data | |
| Formula | C10H12FNO |
| Molar mass | 181.210 g·mol−1 |
Toxicity
Flephedrone has a relatively brief history of human consumption, and there is limited information available regarding its toxicity and potential risks.
Legality
- Denmark prohibited the use of Flephedrone in December 2008.
- It has been illegal since April 2010 in the United Kingdom and Lithuania.
- Brazil has classified Flephedrone as illegal since 2017, considering it an analog or derivative of cathinone.
- Flephedrone is likely illegal in Australia due to its classification as an analog or derivative of cathinone.
- Poland has banned Flephedrone since April 2010.
- Sweden has categorized Flephedrone as a narcotic since October 2010.
- Flephedrone is temporarily listed as a Schedule I controlled substance in the United States. It is permanently listed as a Schedule I controlled substance in Florida, Georgia, and Pennsylvania.
- As of October 2015, 4-FMC is a controlled substance in China.
FAQ
1. What is Flephedrone?
- Flephedrone, or 4-fluoro methcathinone (4-FMC), is a synthetic stimulant drug in the cathinone chemical class.
2. When did Flephedrone emerge as a designer drug?
- Flephedrone gained popularity as a designer drug starting in 2008.
3. What is the legal status of Flephedrone in various countries?
- Flephedrone’s legal status varies by country. It has been illegal in Denmark since December 2008. In the United Kingdom and Lithuania, it became unlawful in April 2010. Brazil classified it as illegal in 2017, considering it an analog or derivative of cathinone. It is likely illegal in Australia for the same reason. Poland banned Flephedrone in April 2010. Sweden has classified it as a narcotic since October 2010. In the United States, Flephedrone is temporarily listed as a Schedule I controlled substance and permanently listed in Florida, Georgia, and Pennsylvania. As of October 2015, 4-FMC (a related sense) is a controlled substance in China.
4. What is known about the safety and toxicity of Flephedrone?
- Flephedrone has a limited history of human use, and its toxicity is not well-established. Due to its classification as a controlled substance in various countries, its use is discouraged and potentially illegal in many places.
5. Are there any other names or synonyms for Flephedrone?
- Flephedrone, also known as 4-FMC, is sometimes referred to by its chemical name, 4-fluoromethcathinone.
Drug regulations and information may change over time, so staying informed about the latest developments and legal status in your region is crucial.
References
- The Brazilian regulatory agency Anvisa, in a resolution issued on July 24, 2023, known as RDC Nº 804, released a list of substances under special control, including narcotic and psychotropic substances. This regulatory update impacts the legal status of certain substances in Brazil.
- In the United States, the Federal Register has implemented temporary measures regarding synthetic cathinones. These measures involve the placement of ten synthetic cathinones into Schedule I of controlled substances. This action reflects efforts to regulate these substances due to potential risks.
- In March 2009, a scientific report authored by Archer RP highlighted a new substance of abuse known as fluoromethcathinone. This substance has garnered attention in the field of forensic science, underscoring the ongoing challenges of identifying and addressing emerging designer drugs.
- An incident involving a bath salt product containing flephedrone and MDPV led to a case of psychosis, as documented in a September 2012 article by Thornton SL et al. This case underscores the dangers associated with these substances and the need for monitoring and regulation.
- Poland introduced legislative changes in April 2011 aimed at combating drug abuse. The “Ustawa z dnia 15 kwietnia 2011” (Law of April 15, 2011) brought amendments to existing anti-drug legislation, reflecting ongoing efforts to address drug-related issues.
- The Swedish Medicines Agency, Läkemedelsverket, maintains a constitutional collection of regulations and guidelines pertaining to medical products and substances. This repository is essential for maintaining and updating pharmaceutical regulations in Sweden.
- The State of Florida issued an emergency rule in response to substances like flephedrone and MDPV being found in products sold as “bath salts.” This regulatory measure aimed to address the risks associated with these substances, particularly in consumer products.
- The General Assembly of Pennsylvania enacted Senate Bill 1006, which aimed to regulate and control certain substances, including synthetic cathinones. Such legislative actions are critical for safeguarding public health and addressing emerging drug threats.
- China’s Food and Drug Administration issued a notice on September 27, 2015, regarding the regulation and control of non-medicinal narcotic drugs and psychotropic substances. This notice reflects China’s efforts to manage and control substances with the potential for misuse.