Beautiful Plants For Your Interior
Summary
CP 55,940, a synthetic cannabinoid, replicates the effects of naturally found THC, one of the active compounds present in cannabis. This compound was originally developed by Pfizer in 1974 but was never brought to the market. Today, it is a valuable research tool for investigating the endocannabinoid system.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 83002-04-4 |
|---|---|
| PubChem CID | 3086156 |
| ChemSpider | 94668 |
| UNII | KFY70972J5 |
| ChEMBL | ChEMBL559612 |
| CompTox Dashboard (EPA) | DTXSID80875516 |
| Chemical and physical data | |
| Formula | C24H40O3 |
| Molar mass | 376.581 g·mol−1 |
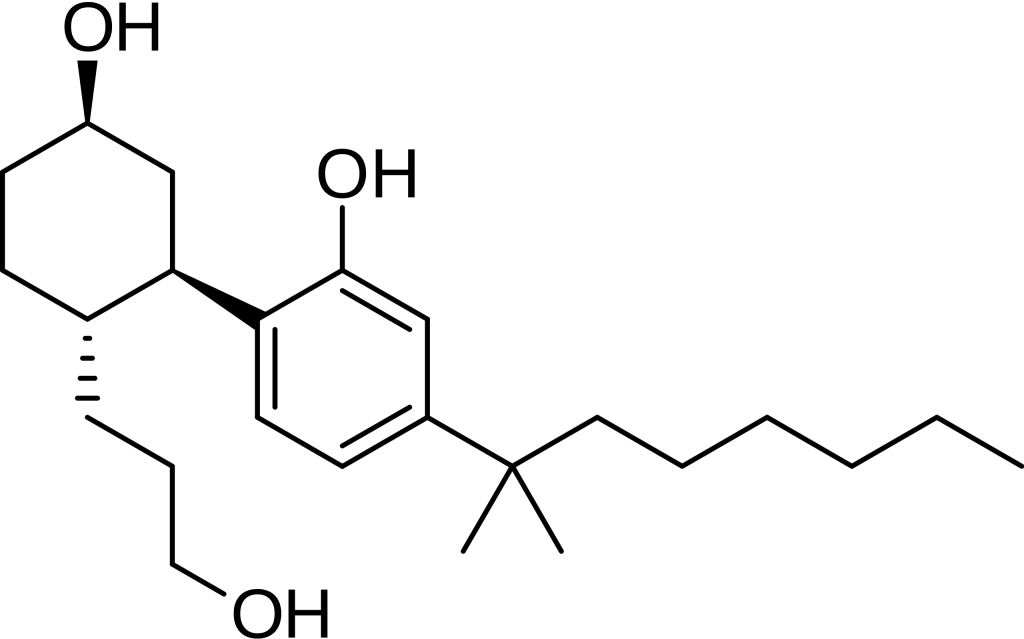
Pharmacology
CP 55,940 exhibits exceptional potency, surpassing Δ9-THC by a factor of 45, and its effects can be completely blocked by rimonabant (SR141716A). It is a full agonist at CB1 and CB2 receptors, boasting Ki values of 0.58 nM and 0.68 nM, respectively. However, it assumes an antagonistic role at GPR55, often called the potential “CB3” receptor. Notably, CP 55,940 binding has been identified in the cytosol of the rat brain’s cerebral cortex. Furthermore, it demonstrates the ability to enhance 5-HT2A receptors in mice.
In vitro studies
CP 55,940 triggered cell apoptosis in hybrid brain cancer cells, specifically the NG 108-15 Mouse neuroblastoma x Rat glioma variety, which involves genetically engineered mouse and rat brain cancer cells.
In vivo studies
CP 55,940 exhibited protective properties on the mitochondria of rat brains when exposed to paraquat.
Additionally, it demonstrated neuroprotective effects by decreasing intracellular calcium release and mitigating hippocampal cell death in cultured neurons exposed to elevated NMDA levels.
FAQ
1. What is CP 55,940?
CP 55,940 is a synthetic cannabinoid compound known for its potent interaction with the endocannabinoid system. It is often used in scientific research to study the effects and mechanisms of cannabinoids.
2. How does CP 55,940 compare to Δ9-THC (delta-9-tetrahydrocannabinol)?
CP 55,940 is approximately 45 times more potent than Δ9-THC, which is one of the primary psychoactive components found in cannabis.
3. What are its receptor interactions?
CP 55,940 is a full agonist at CB1 and CB2 cannabinoid receptors, with specific Ki values of 0.58 nM and 0.68 nM, respectively. It also acts as an antagonist at GPR55, sometimes called the “CB3” receptor.
4. Where is CP 55,940 binding detected?
Binding of CP 55,940 has been observed in the cytosol of the rat brain’s cerebral cortex.
5. Can CP 55,940 influence other receptors or systems?
Yes, it has been noted to upregulate 5-HT2A receptors in mice, indicating potential interactions with serotonin receptor systems.
6. Does CP 55,940 have any medical or therapeutic applications?
CP 55,940 is primarily used as a research tool and has no approved medical applications for human use.
7. Are there any reported side effects or adverse reactions?
The potential side effects or adverse reactions of CP 55,940 are largely unknown, as it is used in controlled laboratory settings and not for human consumption.
8. Can I obtain CP 55,940 for personal use or self-medication?
CP 55,940 is not intended for personal use or self-medication. It is strictly used for scientific research and should not be sought for recreational or therapeutic purposes.
9. Is CP 55,940 legal?
The legal status of CP 55,940 may vary by region and country. It is essential to check your local laws and regulations regarding the possession and use of synthetic cannabinoids.
10. How can I access CP 55,940 for research purposes?
Researchers typically obtain CP 55,940 through authorized suppliers and laboratories specializing in chemical compounds for scientific investigation. Proper licensing and permissions are typically required for its acquisition.
References
- Glass M, Dragunow M, Faull RL (March 1997). “Exploring Cannabinoid Receptors in the Human Brain: A Comprehensive Anatomical and Quantitative Autoradiographic Study across the Fetal, Neonatal, and Adult Human Brain.” Delve into Neuroscience, Volume 77, Issue 2, Pages 299–318. doi:10.1016/s0306-4522(96)00428-9. PMID 9472392.
- Rinaldi-Carmona M, Pialot F, Congy C, Redon E, Barth F, Bachy A, et al. (1996). “Unveiling the Characterization and Distribution of Binding Sites for [3H]-SR 141716A, a Selective Brain (CB1) Cannabinoid Receptor Antagonist, throughout the Rodent Brain.” Unearthed in Life Sciences, Volume 58, Issue 15, Pages 1239–47. doi:10.1016/0024-3205(96)00085-9. PMID 8614277.
- Kapur A, Zhao P, Sharir H, Bai Y, Caron MG, Barak LS, Abood ME (October 2009). “Unconventional Responsiveness of the Orphan Receptor GPR55 to Cannabinoid Ligands.” The Insights within The Journal of Biological Chemistry, Volume 284, Issue 43, Pages 29817–27. doi:10.1074/jbc.M109.050187. PMC 2785612. PMID 19723626.
- Qureshi J, Saady M, Cardounel A, Kalimi M (April 1998). “Discovery and Characterization of a Novel Synthetic Cannabinoid CP 55,940 Binding Partner in Rat Brain Cytosol.” Found in Molecular and Cellular Biochemistry, Volume 181, Issues 1–2, Pages 21–27. doi:10.1023/A:1006855504094. PMID 9562238. S2CID 6200670.
- Franklin JM, Carrasco GA (March 2013). “Stimulating Cannabinoid Receptor Agonists Elevate Serotonin 2A (5-HT(2A)) Receptor Activity via ERK1/2 Signaling.” A Revelation in Synapse, Volume 67, Issue 3, Pages 145–59. doi:10.1002/syn.21626. PMC 3552103. PMID 23151877.
- Tomiyama K, Funada M (November 2011). “Unveiling the Cytotoxicity of Synthetic Cannabinoids Discovered in “Spice” Products: The Involvement of Cannabinoid Receptors and the Caspase Cascade in the NG 108-15 Cell Line.” Discovered in Toxicology Letters, Volume 207, Issue 1, Pages 12–7. doi:10.1016/j.toxlet.2011.08.021. PMID 21907772.
- “General Cell Collection: NG108-15.” An Insightful Resource from Public Health England Culture Collections.
- Velez-Pardo C, Jimenez-Del-Rio M, Lores-Arnaiz S, Bustamante J (September 2010). “Uncovering the Protective Effects of Synthetic Cannabinoids CP55,940 and JWH-015 on Rat Brain Mitochondria when Exposed to Paraquat.” Revealed in Neurochemical Research, Volume 35, Issue 9, Pages 1323–32. doi:10.1007/s11064-010-0188-1. hdl:11336/67604. PMID 20514518. S2CID 821457.
- Zhuang SY, Bridges D, Grigorenko E, McCloud S, Boon A, Hampson RE, Deadwyler SA (June 2005). “Illuminating How Cannabinoids Provide Neuroprotection by Mitigating Intracellular Calcium Release from Ryanodine-Sensitive Stores.” Dissected in Neuropharmacology, Volume 48, Issue 8, Pages 1086–96. doi:10.1016/j.neuropharm.2005.01.005. PMID 15910885. S2CID 14953725.