Beautiful Plants For Your Interior
Summary
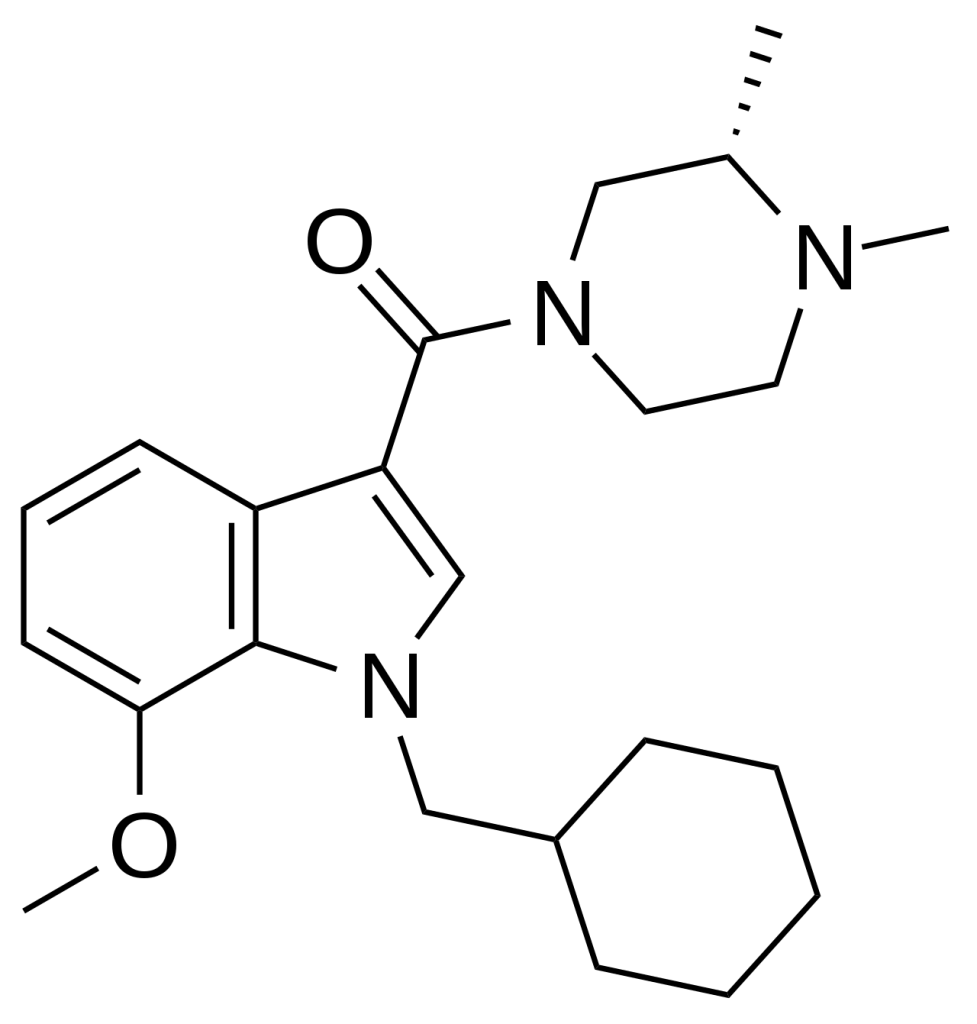
Org 28611 (SCH-900,111) is a pharmaceutical compound created by Organon International, functioning as a potent full agonist at both the CB1 and CB2 cannabinoid receptors. Its development was driven by the quest for a water-soluble cannabinoid agonist intended for intravenous administration as an analgesic. While it effectively met its goal and advanced to Phase II clinical trials in humans, it did not yield significantly favourable results compared to standard drugs (midazolam and morphine) during initial testing.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 639068-99-8 |
|---|---|
| PubChem CID | 10091392 |
| ChemSpider | 8266929 |
| UNII | 1U7V49NTQG |
| ChEMBL | ChEMBL1209709 |
| Chemical and physical data | |
| Formula | C23H33N3O2 |
| Molar mass | 383.536 g·mol−1 |
FAQ
- What is Org 28611 (SCH-900,111)?
- Org 28611, also known as SCH-900,111, is a pharmaceutical compound developed by Organon International. It acts as a potent agonist at the CB1 and CB2 cannabinoid receptors.
- What was the purpose of developing Org 28611?
- The primary goal of developing Org 28611 was to create a water-soluble cannabinoid agonist suitable for intravenous administration as an analgesic.
- Has Org 28611 undergone clinical trials?
- Yes, Org 28611 advanced to Phase II clinical trials in humans. It was evaluated for its potential as both a sedative and an analgesic.
- What were the results of the clinical trials?
- Initial testing of Org 28611 in comparison to standard drugs (midazolam and morphine) did not yield particularly favourable results. The compound’s effectiveness and safety in a clinical setting were assessed.
- Is Org 28611 currently used as a medication?
- Org 28611 is not in everyday medical use, and its development did not progress beyond Phase II clinical trials.
- Are there any potential future applications for Org 28611?
- While Org 28611’s development for analgesic purposes faced challenges, research in cannabinoid receptors continues, and its properties may inspire future results in related areas.
References
- Adam, J. M., et al. (2010). “Design, synthesis, and structure–activity relationships of indole-3-carboxamides as novel water-soluble cannabinoid CB1 receptor agonists.” This study explores the development of water-soluble cannabinoid receptor agonists, including Org 28611, with potential applications in the medicinal field.
- Zuurman, L., Passier, P. C., de Kam, M., Kleijn, H. J., Cohen, A. F., van Gerven, J. M. (August 2009). “Pharmacodynamic and pharmacokinetic effects of the intravenously administered CB1 receptor agonist Org 28611 in healthy male volunteers.” This research delves into the effects of Org 28611 when administered intravenously to human subjects, shedding light on its pharmacological characteristics.
- “A Comparison of Analgesic Efficacy Between a Single Dose of ORG 28611, Morphine, and Placebo After Dental Impaction Surgery.” This clinical study evaluates the analgesic efficacy of Org 28611 in comparison to morphine and a placebo, specifically in the context of dental impaction surgery, providing insights into its potential as a pain-relief medication.