Beautiful Plants For Your Interior
Summary
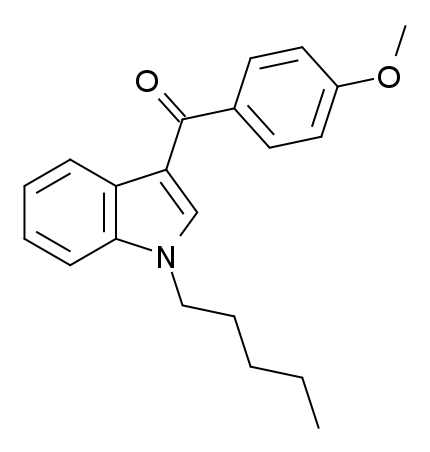
RCS-4, also known as 1-pentyl-3-(4-methoxybenzoyl)indole, is a synthetic cannabinoid compound that has been marketed and sold under various names, including SR-19, BTM-4, Eric-4 (later abbreviated to E-4), and initially, OBT-199.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1345966-78-0 |
|---|---|
| ChemSpider | 24769418 |
| UNII | Y6911BZ2UL |
| CompTox Dashboard (EPA) | DTXSID70158820 |
| ECHA InfoCard | 100.233.383 |
| Chemical and physical data | |
| Formula | C21H23NO2 |
| Molar mass | 321.420 g·mol−1 |
Pharmacology
RCS-4, a potent agonist for cannabinoid receptors, exhibits EC50 values of 146 nM for human CB1 receptors and 46 nM for human CB2 receptors. Methoxyphenyl regioisomers and N-butyl homologues of RCS-4 and its regioisomers also demonstrate vigorous agonist activity at CB1 and CB2 receptors.
Legality
- Sweden prohibited RCS-4 on October 1, 2010, classifying it as a hazardous substance harmful to health. This decision came after its identification as an ingredient in synthetic cannabis products.
- Denmark implemented a ban on RCS-4 on March 11, 2011.
- In August 2011, New Zealand expanded its regulatory measures to include RCS-4 and its 1-butyl homologue, along with the 2-methoxybenzoyl isomers of these compounds. These substances were placed under a temporary class drug schedule, which resembled Class C but was subject to review after 12 months. Personal possession and small-scale use were decriminalized under this regulation, created through the Misuse of Drugs Amendment Act 2011.
- As of October 2015, RCS-4 is designated as a controlled substance in China.
FAQ
- What is RCS-4, and how does it interact with cannabinoid receptors?
- RCS-4 is a synthetic compound known for its potent interaction with cannabinoid receptors. It acts as an agonist, with EC50 values of 146 nM for human CB1 receptors and 46 nM for human CB2 receptors. This means it can bind to and activate these receptors.
- Are there any related compounds with similar properties to RCS-4?
- Yes, methoxyphenyl regioisomers and N-butyl homologues of RCS-4 and its regioisomers also display vigorous agonist activities at CB1 and CB2 receptors. These compounds share similar pharmacological characteristics.
- Why was RCS-4 banned in various countries?
- RCS-4 faced legal restrictions in several countries due to its association with synthetic cannabis products. Its potential health hazards led to its classification as a hazardous substance harmful to health in Sweden and a ban in Denmark. New Zealand also included RCS-4 and related compounds in its temporary class drug schedule, emphasizing the need for regulatory control.
- What is the current legal status of RCS-4 in China?
- As of October 2015, RCS-4 is considered a controlled substance in China, subject to legal restrictions and regulations.
- Is RCS-4 used for medicinal or recreational purposes?
- RCS-4 is primarily known for its role in research and has not been approved for medical or recreational use. It is crucial to adhere to legal and safety guidelines when working with such compounds.
- Where can I find more information about RCS-4 and its effects on cannabinoid receptors?
- For in-depth information on RCS-4 and its interactions with cannabinoid receptors, consider referring to scientific publications, academic resources, and consulting experts in cannabinoid pharmacology. Always prioritize safety and compliance with relevant regulations when handling such substances.
References
- The “Ustawa z dnia 15 kwietnia 2011 r. o zmianie ustawy o przeciwdziałaniu narkomanii” (Law of April 15, 2011, amending the Law on Counteracting Drug Addiction) is a legislative document from Poland. It was published in the “Dziennik Ustaw” (Journal of Laws) in 2011 under number 105, entry 614. This law introduced changes related to drug addiction prevention and control.
- Banister SD, Stuart J, Conroy T, and colleagues conducted research in 2015, exploring the “Structure–activity relationships of synthetic cannabinoid designer drug RCS-4 and its regioisomers and C4 homologues.” This study, published in “Forensic Toxicology,” investigated the pharmacological properties and relationships of RCS-4 and its related compounds, shedding light on their effects.
- The “Swedish Code of Statutes Regulation (2010:1086)” played a crucial role in regulating substances in Sweden. This regulation, numbered 1086 in 2010, was a legal framework for controlling various substances, including those with potential health risks.
- A supplementary document to the “Swedish Code of Statutes Regulation (2010:1086)” provided additional information and guidance. It is part of the legal framework for substance control in Sweden.
- “Bilag 1 – Liste over euforiserende midler omfattet af bekendtgørelsen” (Appendix 1 – List of euphoric agents covered by the announcement) is associated with the Ministry of Health and Prevention. This document includes a list of substances with euphoric properties and falls under specific regulations in Denmark.
- In August 2011, New Zealand passed legislation regarding synthetic cannabis products. This action was reported by “The New Zealand Herald” in an article titled “Kronic ban passed by Parliament,” highlighting the decision to regulate these substances.
- The availability of synthetic cannabis products in New Zealand was affected by the legislative changes. “The New Zealand Herald” reported on August 9, 2011, that “Synthetic cannabis [was] off shelves by Wednesday,” indicating the prompt implementation of the new regulations.
- New Zealand Gazette, in its issue dated August 9, 2011, published Departmental Notices related to the Misuse of Drugs Act 1975. These notices, specifically the Temporary Class Drug Notice, were instrumental in controlling the distribution and use of certain substances.
- On September 27, 2015, the China Food and Drug Administration (CFDA) issued a notice in Chinese titled “关于印发《非药用类麻醉药品和精神药品列管办法》的通知” (Notice on the issuance of the “Regulations for the Supervision and Administration of Non-Medical Narcotic Drugs and Psychotropic Substances”). This notice outlined regulations for the supervision and control of non-medical narcotics and psychotropic substances in China.