Beautiful Plants For Your Interior
Summary
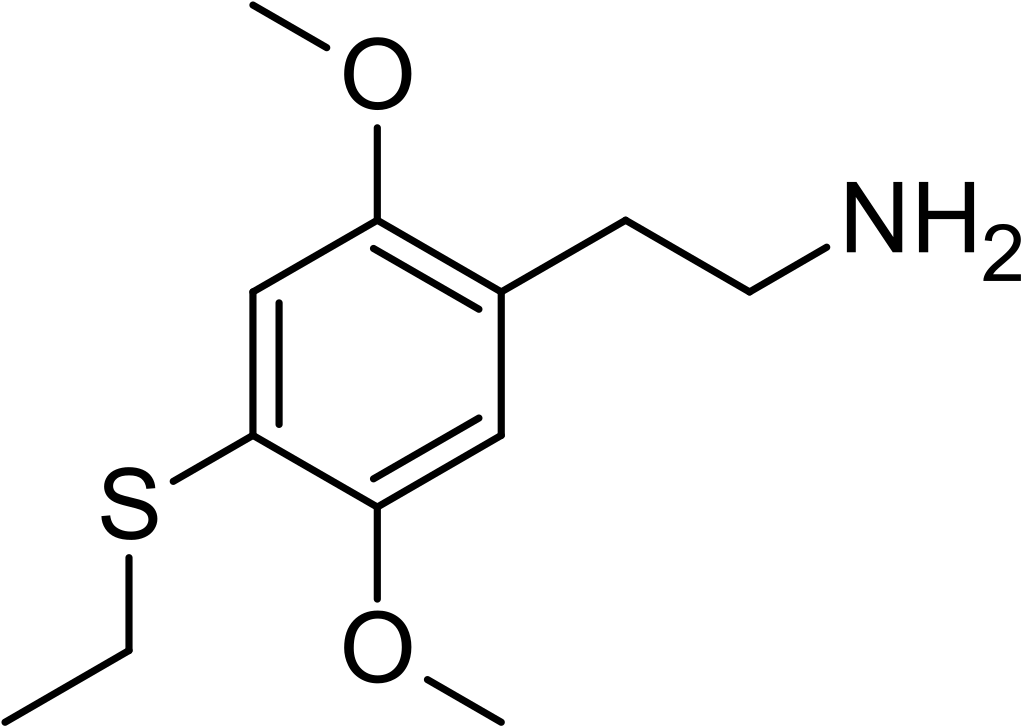
2C-T-2 belongs to the 2C family and is a phenethylamine known for its psychedelic and entactogenic properties. This compound was initially synthesized by Alexander Shulgin in 1981 and earned its place among the “magical half-dozen” due to its significant role in the realm of psychedelic phenethylamines. 2C-T-2 shares similarities in both structure and pharmacodynamics with another compound, 2C-T-7, commonly referred to as “Blue Mystic.”
| Identifiers | |
|---|---|
| CAS Number | 207740-24-7 |
| 3D model (JSmol) | Interactive image |
| ChEMBL | ChEMBL339223 |
| ChemSpider | 16787961 |
| ECHA InfoCard | 100.241.509 |
| PubChemCID | 12074193 |
| UNII | WPS2KSX2TJ |
| CompTox Dashboard(EPA) | DTXSID40174850 |
Pharmacology
2C-T-2’s hallucinogenic and entheogenic effects are believed to be primarily attributed to its actions as an agonist on serotonin receptors, specifically the 5-HT2A, 5-HT2B, and 5-HT2C receptors. This mechanism of action is shared with other hallucinogenic compounds, including tryptamines and phenethylamines, albeit to varying degrees. Additionally, 2C-T-2 has been found to act as a partial agonist on adrenergic receptors.
Dangers
The use of 2C-T-2, as well as other compounds in the 2C chemical series, carries a potential risk of neurotoxicity, affecting neurons containing serotonin and dopamine. This risk is further exacerbated when 2C series drugs are combined with substances such as alcohol, MDMA, and methamphetamine, particularly in serotonergic-containing cells.
Severe intoxication resulting from 2C series drug use may manifest as intense hallucinations, heightened agitation, aggressive behaviour, violence, dysphoria, elevated blood pressure, rapid heart rate (tachycardia), seizures, and increased body temperature (hyperthermia).
Drug prohibition laws
Argentina
2C-T-2 is classified as a controlled substance in Argentina, along with 2C-B and 2C-I.
Canada
As of October 31, 2016, 2C-T-2 is a controlled substance listed under Schedule III in Canada.
China
2C-T-2 is designated as a controlled substance in China, effective as of October 2015.
Netherlands
The Netherlands holds the distinction of being the first country to prohibit and categorize 2C-T-2 as a hard drug through legislation. In April 1999, 2C-T-2 was included in List I of controlled substances under the Opium Law.
Sweden
2C-T-2 is categorized as Schedule I in Sweden. It was initially labelled as a “health hazard” under the Act on the Prohibition of Certain Goods Dangerous to Health, commencing on April 1, 1999, per SFS 1999:58. This classification rendered it illegal to sell or possess.
Additionally, the Riksdag added 2C-T-2 to the Narcotic Drugs Punishments Act, classifying it under Swedish Schedule I, which pertains to substances, plant materials, and fungi that typically lack medical applications. This came into effect on March 16, 2004, as published by the Medical Products Agency (MPA) in regulation LVFS 2004:3, listing it as 2C-T-2, 2,5-dimethoxy-4-ethylthiophenethylamine.
United Kingdom
2C-T-2, along with all other compounds featured in PiHKAL, is illegal in the United Kingdom.
United States
In the United States, 2C-T-2 is explicitly classified as a Schedule I substance under SEC. 1152 of S.3187: Food and Drug Administration Safety and Innovation Act of 2012.
Australia
Australia designates 2C-T-2 as a Schedule 9 prohibited substance under the Poisons Standard, dated October 2015. A Schedule 9 substance is one prone to abuse or misuse, and its manufacture, possession, sale, or use is typically prohibited by law. Exceptions may apply for medical or scientific research, as well as for analytical, teaching, or training purposes, with approval from Commonwealth and State or Territory Health Authorities.
FAQ
1. What is 2C-T-2?
2C-T-2 is a psychedelic and entactogenic phenethylamine. It belongs to the 2C family of compounds known for their psychoactive effects.
2. How was 2C-T-2 discovered?
2C-T-2 was first synthesized in 1981 by Alexander Shulgin, a renowned chemist and pharmacologist. He considered it one of the “magical half-dozen,” which includes significant psychedelic compounds.
3. What are its effects?
2C-T-2 produces hallucinogenic and entheogenic effects characterized by visual and sensory alterations. It interacts with serotonin receptors, primarily 5-HT2A, 5-HT2B, and 5-HT2C.
4. Is 2C-T-2 legal?
The legal status of 2C-T-2 varies by country. It is illegal in many places, including the United States, the United Kingdom, Canada, and several European countries. It is essential to check your local laws and regulations.
5. Are there safety concerns with 2C-T-2?
The use of 2C-T-2 may carry risks, including potential neurotoxicity. There have been reports of severe intoxication when used with other substances like alcohol, MDMA, and methamphetamine. It’s crucial to use caution and be aware of potential risks.
6. Can I use 2C-T-2 for medical or therapeutic purposes?
2C-T-2 is not approved for medical use, and its recreational use is illegal in many places. Research on its therapeutic potential is limited.
7. What precautions should I take if I plan to use 2C-T-2?
If you are in an area where 2C-T-2 is legal, consider harm reduction strategies, such as starting with a low dose, using it in a safe and controlled environment, and having a trusted person present. Always be aware of the legal status in your location.
8. Are there any reported cases of 2C-T-2-related issues?
There have been reports of incidents related to 2C-T-2, including hospitalizations. It’s essential to stay informed about potential risks and effects.
9. Where can I find more information about 2C-T-2?
You can explore resources such as books by Alexander Shulgin, scientific articles, and online forums to learn more about 2C-T-2. However, ensure that you prioritize your safety and adhere to local laws.
10. What should I do if I have questions about 2C-T-2?
If you have specific questions or concerns about 2C-T-2, consult with a healthcare professional or seek guidance from experts in the field of psychedelic substances. Always prioritize safety and responsible use.
References
- In September 2005, Theobald DS and colleagues conducted a study published in the Journal of Mass Spectrometry, titled “New designer drug 2,5-dimethoxy-4-ethylthio-beta-phenethylamine (2C-T-2): studies on its metabolism and toxicological detection in rat urine using gas chromatography/mass spectrometry.” The study delved into the metabolic pathways and toxicological detection of 2C-T-2 in rat urine through gas chromatography/mass spectrometry.
- For further insights into 2C-T-2, you can explore “#40 2C-T-2” in Erowid Online Books’ “PIHKAL” resources.
- In December 2015, Rickli A and his team published “Receptor interaction profiles of novel N-2-methoxybenzyl (NBOMe) derivatives of 2,5-dimethoxy-substituted phenethylamines (2C drugs)” in Neuropharmacology. This research focused on the receptor interaction profiles of NBOMe derivatives, including 2C-T-2, and their effects on phenethylamines.
- Eshleman AJ, Forster MJ, Wolfrum KM, Johnson RA, Janowsky A, Gatch MB presented the study “Behavioral and neurochemical pharmacology of six psychoactive substituted phenethylamines: mouse locomotion, rat drug discrimination and in vitro receptor and transporter binding and function” in the journal Psychopharmacology (March 2014). The study covered topics such as mouse locomotion, rat drug discrimination, and in vitro receptor and transporter interactions of these psychoactive phenethylamines.
- In August 2016, Rickli A and his team published a study titled “Receptor interaction profiles of novel psychoactive tryptamines compared with classic hallucinogens” in the European Neuropsychopharmacology journal. This study compared receptor interaction profiles of novel psychoactive tryptamines with classic hallucinogens to provide insights into their effects.
- In May 2018, Luethi D, Trachsel D, Hoener MC, and Liechti ME delved into “Monoamine receptor interaction profiles of 4-thio-substituted phenethylamines (2C-T drugs)” in Neuropharmacology. This research explored the monoamine receptor interaction profiles of 4-thio-substituted phenethylamines, which include 2C-T-2, and their potential effects.
- In a study published in November 2019 in the Archives of Toxicology, Dai and his team investigated the neurotoxicity of T-2 toxin, focusing on the role of oxidative stress and mitochondrial dysfunction in this context.
- Asanuma M, Miyazaki I, and Funada M published research in July 2020 in Forensic Toxicology. Their work examined the neurotoxic potential of psychoactive phenethylamines in the “2C series” using cultured monoaminergic neuronal cell lines.
- Dean BV, Stellpflug SJ, Burnett AM, and Engebretsen KM provided a comprehensive review in June 2013 in the Journal of Medical Toxicology, titled “2C or not 2C: phenethylamine designer drug review.” The review discussed phenethylamine designer drugs, their pharmacological properties, and potential risks.
- In Argentina, “DECRETO 299/2010” is a legal document from the NATIONAL EXECUTIVE POWER (P.E.N.), addressing the regulation of narcotic substances and chemical compounds within the scope of Law 23,737, with a focus on the inclusion of 2C-T-2 in the list of controlled substances.
- Canada Gazette – Regulations Amending the Food and Drug Regulations provides information on regulations amending the Food and Drug Regulations in Canada, particularly regarding 2C-phenethylamines, which include 2C-T-2.
- In China, the “关于印发《非药用类麻醉药品和精神药品列管办法》” notice from the China Food and Drug Administration outlines regulations related to non-medicinal psychoactive substances, including 2C-T-2.
- In Sweden, the “Förordning (1999:58) om förbud mot vissa hälsofarliga varor” regulation addresses the prohibition of certain health-hazardous goods, including 2C-T-2, and the legal restrictions surrounding its sale and possession.
- The “Läkemedelsverkets författningssamling” in Sweden, published by the Medical Products Agency, lists 2C-T-2 under Schedule I, which encompasses substances without recognized medical use.
- “21 U.S. Code § 812” outlines the schedules of controlled substances in the United States, including the classification of 2C-T-2.
- In Australia, the “Poisons Standard October 2015” from the Federal Register of Legislation provides information on the scheduling of 2C-T-2 as a prohibited substance under the Poisons Standard, outlining its potential uses and restrictions.