Beautiful Plants For Your Interior
Summary
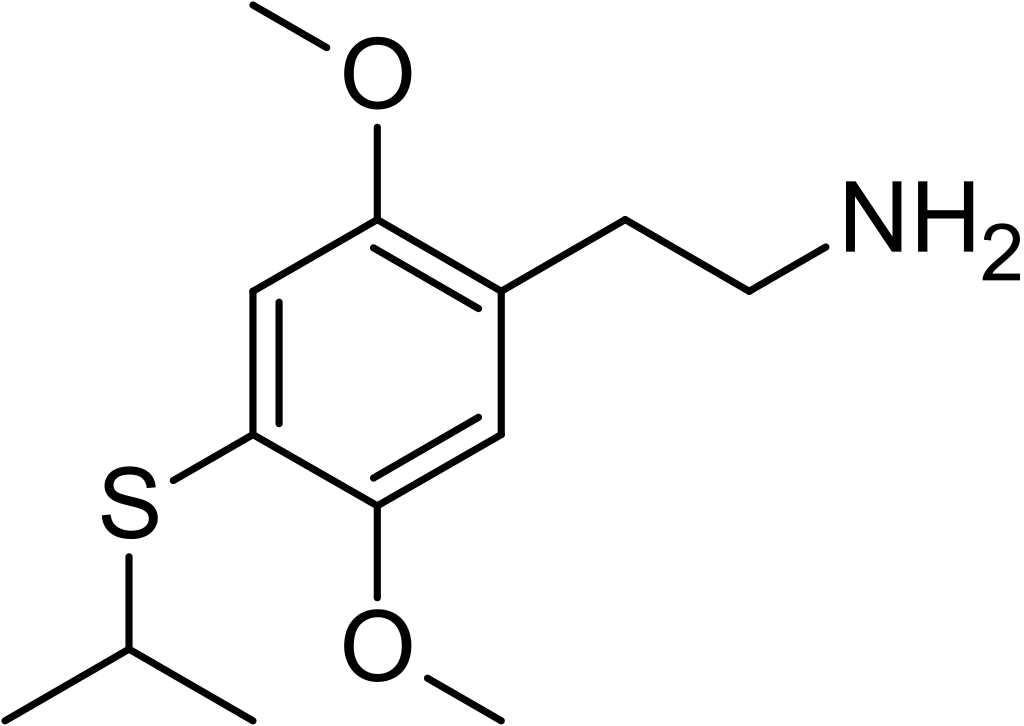
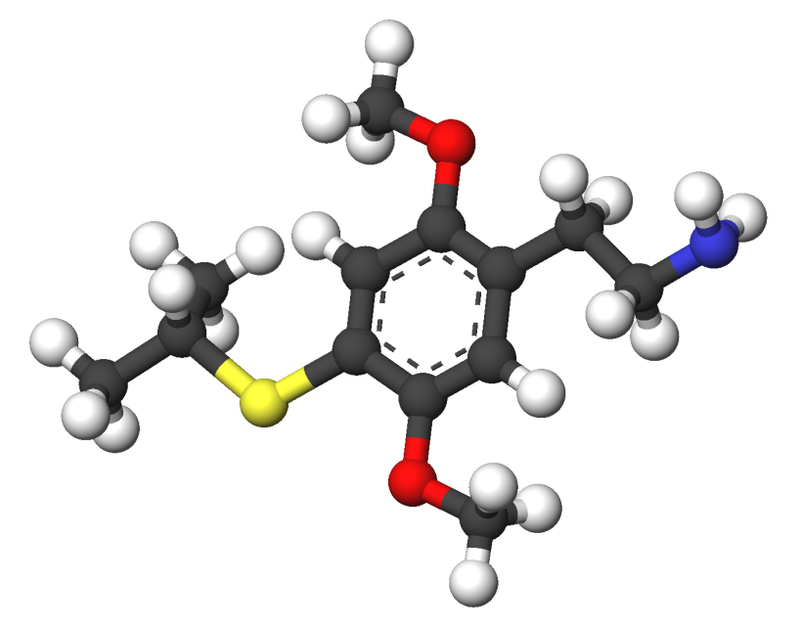
2C-T-4, scientifically known as 2,5-dimethoxy-4-isopropylthiophenethylamine, belongs to the 2C family of psychedelic phenethylamines. Renowned chemist Alexander Shulgin was the first to synthesize this compound. It is primarily utilized as a recreational drug with entheogenic properties.
| Identifiers | |
|---|---|
| CAS Number | 207740-25-8 [chemspider] |
| 3D model (JSmol) | Interactive image |
| ChEMBL | ChEMBL338259 |
| ChemSpider | 21106232 |
| PubChemCID | 44350070 |
| UNII | 558WSD71D4 |
| CompTox Dashboard(EPA) | DTXSID60893915 |
Chemistry
2C-T-4 is the structural counterpart of aleph-4, with a full chemical designation as 2-[4-(isopropylthio)-2,5-dimethoxyphenyl]-ethanamine. This compound shares structural and pharmacodynamic characteristics with 2C-T-7 and 2C-T-19.
Effects
2C-T-4 induces gradual psychedelic and entheogenic effects, which may extend for a duration of 8 to 16 hours, although these claims require medical citation for validation. After ingestion, some users may encounter a delay of up to an hour before experiencing any noticeable effects. The responses to 2C-T-4 can be highly variable among individuals, encompassing hallucinations, euphoria, and, in some cases, intense discomfort and anxiety. Alexander Shulgin dedicated a chapter in the initial section of his book PiHKAL to this compound, recounting a profound “plus-four” psychedelic experience brought about by a twelve-milligram dosage.
Pharmacology
The precise mechanism underlying the hallucinogenic and entheogenic effects of 2C-T-4 remains to be definitively elucidated. Nonetheless, these effects are probably a consequence of its activity as a serotonin receptor agonist, primarily at the 5-HT2A receptor in the brain. This mechanism of action is shared by various hallucinogenic tryptamines and phenethylamines, for which the mechanism of action has been ascertained.
Drug prohibition laws
Canada
Effective from October 31, 2016, 2C-T-4 is classified as a controlled substance (Schedule III) in Canada.
China
Since October 2015, 2C-T-4 has been designated as a controlled substance in China.
Denmark
2C-T-4 has been included in the list of Schedule B controlled substances in Denmark.
Sweden
In Sweden, the health ministry, Statens folkhälsoinstitut, categorized 2C-T-4 as a “health hazard” under the Act on the Prohibition of Certain Goods Dangerous to Health (Lagen om förbud mot vissa hälsofarliga varor) on July 15, 2007. This classification, as per regulation SFS 2007:600, identifies it as 2,5-dimetoxi-4-isopropyltiofenetylamin (2C-T-4), rendering both its sale and possession illegal.
United States
Since July 9, 2012, 2C-T-4 has been designated as a Schedule I substance in the United States under the Synthetic Drug Abuse Prevention Act of 2012.
Homologue
2C-T-4 possesses a structural isomer called Ψ-2C-T-4 (2,6-dimethoxy-4-(i)-propylthiophenethylamine). Alexander Shulgin experimented with a 12 mg dose of this compound.
At this dose, the effects were notably brief and limited. Nevertheless, considering the research on the more extensively studied compound Ψ-DOM, it is plausible that Ψ-2C-T-4’s potency is approximately one-third that of 2C-T-4 itself. Therefore, a more effective dose of Ψ-2C-T-4 may fall within the range of 20–60 mg; however, it’s crucial to exercise extreme caution when contemplating higher doses, as they may lead to potential toxic side effects.
FAQ
- What is 2C-T-4?
- 2C-T-4 is a psychedelic phenethylamine compound that belongs to the 2C family. It is known for its hallucinogenic and entheogenic effects when ingested.
- Who synthesized 2C-T-4?
- 2C-T-4 was first synthesized by Alexander Shulgin, a renowned chemist and pharmacologist who researched and documented various psychoactive substances.
- How does 2C-T-4 affect users?
- The effects of 2C-T-4 are known to develop slowly and can last for a significant duration, ranging from 8 to 16 hours. However, the specific effects can vary significantly among individuals, encompassing everything from hallucinations and euphoria to intense sickness and anxiety.
- Is 2C-T-4 featured in Alexander Shulgin’s book, PiHKAL?
- Yes, 2C-T-4 is featured in the first part of Alexander Shulgin’s book, PiHKAL (Phenethylamines I Have Known And Loved). Shulgin described his own experience with a 12 mg dose, noting it as an intense “plus-four” psychedelic encounter.
- What is the mechanism of action for 2C-T-4’s effects?
- The precise mechanism of action for 2C-T-4’s hallucinogenic and entheogenic effects has yet to be conclusively established. However, it likely acts as a 5-HT2A serotonin receptor agonist in the brain, similar to the mechanism shared by other known hallucinogenic compounds in the tryptamine and phenethylamine classes.
- Is 2C-T-4 legal?
- The legal status of 2C-T-4 varies by country. As of 2012, in the United States, it is classified as a Schedule I substance, making it illegal to possess, distribute, or manufacture. In other countries, such as Canada and China, it is also considered a controlled substance. Be sure to check your local laws and regulations.
- Are there any related compounds to 2C-T-4?
- Yes, there is a structural isomer of 2C-T-4 called Ψ-2C-T-4 (2,6-dimethoxy-4-(i)-propylthiophenethylamine), which has been studied. It is believed to have lower potency compared to 2C-T-4.
- What is the recommended dosage for 2C-T-4?
- The ideal dosage for 2C-T-4 can vary, but caution is advised. While Shulgin’s experience was based on a 12 mg dose, some research suggests that Ψ-2C-T-4, a related compound, might be effective in the range of 20–60 mg. It’s essential to approach dosage with care and awareness of potential side effects.
- What precautions should be taken when using 2C-T-4?
- Safety is paramount when dealing with psychoactive substances. It’s advisable to be well-informed, start with lower doses, and have a trusted and sober individual present as a guide or sitter. Ensure you are in a safe and comfortable environment, and avoid using substances with unknown interactions.
- Is there a risk associated with high doses of 2C-T-4?
- High doses of 2C-T-4 may carry the risk of toxic side effects. Therefore, it’s essential to adhere to responsible and moderate dosing practices and seek medical attention if any adverse effects occur.
References
- Shulgin, Alexander; Shulgin, Ann (September 1991). PiHKAL: A Chemical Love Story. Berkeley, California: Transform Press. ISBN 0-9630096-0-5. OCLC 25627628.
- “Canada Gazette – Regulations Amending the Food and Drug Regulations (Part J — 2C-phenethylamines)”. 4 May 2016.
- “关于印发《非药用类麻醉药品和精神药品列管办法》的通知” (in Chinese). China Food and Drug Administration. 27 September 2015. Archived from the original on 1 October 2015. Retrieved 1 October 2015.
- “Retsinformation”.
- Larsson, Maria. “Svensk författningssamling” (PDF). Retrieved January 24, 2022.
- Portman. “Synthetic Drug Abuse Prevention Act of 2012”. Govtrack. Retrieved 22 July 2012.