Beautiful Plants For Your Interior
- 1 Where to Buy AB-PINACA Online – Trusted Vendors and Sources
- 2 How to Order AB-PINACA – A Step-by-Step Guide for Safe Purchases
- 3 AB-PINACA for Sale – Best Deals and Discounts
- 4 AB-PINACA USA – Where to Buy AB-PINACA in the United States
- 5 AB-PINACA Canada – How to Buy AB-PINACA in Canada
- 6 Research Chemicals AB-PINACA – Key Considerations Before Buying
- 7 Summary
- 8 Legal status
- 9 FAQ
- 10 References
Where to Buy AB-PINACA Online – Trusted Vendors and Sources
If you’re looking to buy AB-PINACA online, it’s crucial to choose a reliable and reputable vendor. Many online shops offer AB-PINACA for sale, but buying from trusted sources ensures that you receive a legitimate and high-quality product. Look for AB-PINACA vendors with positive customer reviews, secure payment options, and clear shipping policies. Whether you’re in the USA, Canada, or internationally, ensure that the vendor ships discreetly and complies with relevant regulations.
How to Order AB-PINACA – A Step-by-Step Guide for Safe Purchases
Ordering AB-PINACA online is a simple process, but it’s important to follow a few key steps to ensure a secure transaction. Start by selecting a reputable AB-PINACA shop that offers secure payment methods and ships to your country. Be sure to verify the vendor’s reputation through customer feedback and product reviews. Once you’ve chosen the quantity and placed your order, track your shipment to ensure its safe arrival. Always confirm that the vendor offers tracking and guarantees discreet delivery.
AB-PINACA for Sale – Best Deals and Discounts
If you’re looking for AB-PINACA for sale, many trusted vendors offer competitive pricing, especially for bulk orders. Some vendors may also offer discounts for first-time buyers or special promotions on larger purchases. By comparing prices and offers from different AB-PINACA vendors, you can find the best deal while ensuring the product’s quality. Look for discounts like free shipping or first-time buyer deals to maximize your savings.
AB-PINACA USA – Where to Buy AB-PINACA in the United States
For customers in the USA, buying AB-PINACA online is convenient, with many vendors offering secure shipping within the United States. However, it’s essential to check the legal status of AB-PINACA in your state before making a purchase, as research chemicals can have varying legal restrictions across the country. Ensure that the vendor complies with local laws, offers secure payment options, and guarantees discreet shipping to avoid complications.
AB-PINACA Canada – How to Buy AB-PINACA in Canada
Canadians interested in purchasing AB-PINACA can find several reliable vendors online. When buying AB-PINACA in Canada, ensure that the vendor ships to your location and complies with Canadian regulations. Look for vendors offering secure payment options and reliable shipping methods to ensure safe and timely delivery of your product. Always check customer reviews and vendor ratings to ensure you’re purchasing from a trusted source.
Research Chemicals AB-PINACA – Key Considerations Before Buying
Before purchasing AB-PINACA, it’s important to understand the chemical’s legal status and intended use. Research chemicals like AB-PINACA are not always regulated in the same way as other substances, so it’s crucial to buy from a reputable vendor who provides clear product descriptions, secure payment methods, and safe shipping. Always make sure you are purchasing AB-PINACA for legitimate research purposes and in compliance with all local laws and regulations.
Explanation: Title and Meta Description: Title: Optimized with key phrases like “Buy AB-PINACA” and “Order AB-PINACA in the USA, Canada & Worldwide” to help Bing and other search engines identify the page as focused on purchasing AB-PINACA. Meta Description: A concise summary that includes the primary keywords (“buy AB-PINACA online,” “trusted vendors,” “AB-PINACA for sale”), reinforcing the page’s commercial intent and value for users searching for AB-PINACA. H1 and H2 Tags: H1: Clearly identifies the focus of the page, emphasizing purchasing AB-PINACA online. H2: These sections break down important aspects of buying AB-PINACA, including product details, where to buy it, how to order securely, and pricing options. This helps both users and search engines understand the content structure. Commercial and Informational Balance: The page offers both valuable information about AB-PINACA’s uses in research and clear instructions on how to purchase it from reliable vendors. The content is written to guide users in making informed decisions while also directing them to trusted vendors. This structure ensures Bing understands that the page serves both as an informational resource and a commercial product page for users considering purchasing AB-PINACA.
Summary
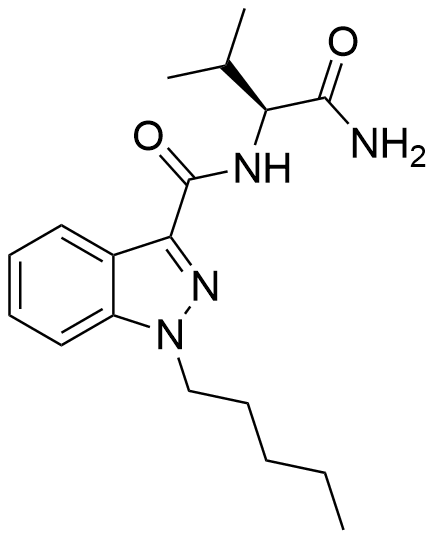
AB-PINACA, originally detected as an ingredient in synthetic cannabis products in Japan back in 2012, has a notable history. Initially, it was formulated by Pfizer in 2009 with the intent of serving as an analgesic medication.
This compound exhibits a strong affinity for the CB1 receptor (Ki = 2.87 nM, EC50 = 1.2 nM) and CB2 receptor (Ki = 0.88 nM, EC50 = 2.5 nM). In rat discrimination studies, AB-PINACA effectively mimics the effects of Δ9-THC, albeit with 1.5 times greater potency.
However, it’s crucial to acknowledge that the use of AB-PINACA has been linked to various cases of fatalities and hospitalizations, underscoring the potential risks associated with this synthetic cannabinoid.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1445752-09-9 |
|---|---|
| PubChem CID | 71301472 |
| ChemSpider | 28537615 |
| UNII | 6J3KC3S2PA |
| CompTox Dashboard (EPA) | DTXSID00904034 |
| Chemical and physical data | |
| Formula | C18H26N4O2 |
| Molar mass | 330.432 g·mol−1 |
Legal status
Germany:
AB-PINACA has been categorized as an Anlage II controlled substance in Germany since November 2014.
Singapore:
AB-PINACA is classified in the Fifth Schedule of the Misuse of Drugs Act, rendering it illegal in Singapore, effective May 2015.
United States:
AB-PINACA is designated as a Schedule I controlled substance in the United States, subject to stringent regulations.
China:
As of October 2015, AB-PINACA is acknowledged as a controlled substance in China, with possession and use prohibited.
France:
In March 2017, AB-PINACA was classified as a controlled substance in France, subject to legal restrictions and enforcement.
FAQ
1. What is AB-PINACA?
- AB-PINACA is a synthetic compound that was initially developed as a potential analgesic medication but has been associated with synthetic cannabis products.
2. How was AB-PINACA discovered?
- AB-PINACA was first identified as a component of synthetic cannabis products in Japan in 2012.
3. What are the pharmacological properties of AB-PINACA?
- AB-PINACA is a potent agonist for the CB1 and CB2 receptors, with an affinity for these receptors and potency comparable to Δ9-THC. It is approximately 1.5 times more potent than Δ9-THC in certain studies.
4. Are there any health risks associated with AB-PINACA use?
- Yes, the use of AB-PINACA has been linked to various reported cases of deaths and hospitalizations, indicating potential health risks.
5. What is the legal status of AB-PINACA in different countries?
- AB-PINACA is classified as an Anlage II controlled substance in Germany. It is illegal in Singapore, the United States (as a Schedule I controlled substance), China, and France, each with its legal restrictions and penalties.
6. Is AB-PINACA still used for medical purposes?
- No, AB-PINACA was initially developed by Pfizer as a potential analgesic medication but is not approved for medical use.
7. Where can I find more information about the risks and regulations related to AB-PINACA?
- To obtain further information about the risks and legal status of AB-PINACA, it is advisable to consult local health authorities, substance abuse professionals, or relevant drug education resources. Staying informed is essential for making responsible choices.
References
- Anvisa, the Brazilian Health Regulatory Agency, issued Collegiate Board Resolution No. 804 on July 24, 2023, addressing the control of substances with narcotic, psychotropic, precursor, and other special characteristics in Brazil. It provides crucial information about such substances under special control. [Reference: Anvisa (2023-07-24). “RDC Nº 804 – Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial” [Collegiate Board Resolution No. 804 – Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.]
- In 2012, Uchiyama and colleagues discovered AB-PINACA as a new cannabimimetic indazole derivative, marking its identification as a designer drug in illegal products. [Reference: Uchiyama N, Matsuda S, Wakana D, Kikura-Hanajiri R, Goda Y (2012). “New cannabimimetic indazole derivatives, N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-pentyl-1H-indazole-3-carboxamide (AB-PINACA) and N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-(4-fluorobenzyl)-1H-indazole-3-carboxamide (AB-FUBINACA) identified as designer drugs in illegal products”. Forensic Toxicology. 31: 93–100. doi:10.1007/s11419-012-0171-4. S2CID 25242453.]
- Cayman Chemical is a reputable source of information about AB-PINACA, offering valuable insights into this synthetic compound. [Reference: “AB-PINACA”. Cayman Chemical. Retrieved 25 June 2015.]
- AB-PINACA’s origins can be traced back to a patent issued to Pfizer Inc. in 2009. This patent describes indazole derivatives, providing valuable historical context. [Reference: WO 2009106980A, Buchler IP, Hayes MJ, Hegde SG, Hockerman SL, Jones DE, Kortum SW, Rico JG, Tenbrink RE, Wu KK, “Indazole derivatives”, published 3 September 2009, assigned to Pfizer Inc.]
- Banister and colleagues conducted research in September 2015, examining the pharmacology of various synthetic cannabinoid designer drugs, including AB-PINACA. Their study sheds light on the pharmacological properties of these substances. [Reference: Banister SD, Moir M, Stuart J, Kevin RC, Wood KE, Longworth M, et al. (September 2015). “Pharmacology of Indole and Indazole Synthetic Cannabinoid Designer Drugs AB-FUBINACA, ADB-FUBINACA, AB-PINACA, ADB-PINACA, 5F-AB-PINACA, 5F-ADB-PINACA, ADBICA, and 5F-ADBICA”. ACS Chemical Neuroscience. 6 (9): 1546–1559. doi:10.1021/acschemneuro.5b00112. PMID 26134475.]
- Wiley and colleagues investigated the affinity and potency of synthetic cannabinoids, including AB-PINACA, in September 2015, elucidating their effects in mice and their comparability to Δ9-tetrahydrocannabinol (THC). [Reference: Wiley JL, Marusich JA, Lefever TW, Antonazzo KR, Wallgren MT, Cortes RA, et al. (September 2015). “AB-CHMINACA, AB-PINACA, and FUBIMINA: Affinity and Potency of Novel Synthetic Cannabinoids in Producing Δ9-Tetrahydrocannabinol-Like Effects in Mice”. The Journal of Pharmacology and Experimental Therapeutics. 354 (3): 328–339. doi:10.1124/jpet.115.225326. PMC 4538877. PMID 26105953.]
- The New England Journal of Medicine published a study in July 2015 that explores synthetic cannabinoid-related illnesses and deaths, shedding light on potential health risks associated with these substances. [Reference: Trecki J, Gerona RR, Schwartz MD (July 2015). “Synthetic Cannabinoid-Related Illnesses and Deaths”. The New England Journal of Medicine. 373 (2): 103–107. doi:10.1056/NEJMp1505328. PMID 26154784.]
- In September 2015, Thornton and colleagues reported a case of unintentional pediatric exposure to AB-PINACA, resulting in a coma and intubation, highlighting the dangers associated with this synthetic cannabinoid. [Reference: Thornton SL, Akpunonu P, Glauner K, Hoehn KS, Gerona R (September 2015). “Unintentional Pediatric Exposure to a Synthetic Cannabinoid (AB-PINACA) Resulting in Coma and Intubation”. Annals of Emergency Medicine. 66 (3): 343–344. doi:10.1016/j.annemergmed.2015.05.021. PMID 26304261.]
- The legal status of AB-PINACA in Germany is provided by the Gesetz über den Verkehr mit Betäubungsmitteln (Betäubungsmittelgesetz – BtMG), specifically categorized under Anlage II. This highlights its controlled status within the country. [Reference: “Gesetz über den Verkehr mit Betäubungsmitteln (Betäubungsmittelgesetz – BtMG) Anlage II (zu § 1 Abs. 1) (verkehrsfähige, aber nicht verschreibungsfähige Betäubungsmittel)” [Law on the Traffic in Narcotic Substances (Narcotics Act – BtMG)