Beautiful Plants For Your Interior
Summary
ADB-BINACA is a synthetic cannabinoid designer drug that has been identified as a component in certain synthetic cannabis formulations. Initially conceived by Pfizer for its potential analgesic properties, this compound is a robust agonist for the CB1 receptor, boasting an impressive binding affinity (Ki) of 0.33 nM and an EC50 of 14.7 nM.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1185282-27-2 |
|---|---|
| PubChem CID | 129406620 |
| ChemSpider | 57621565 |
| UNII | PUVP61SOSI |
| Chemical and physical data | |
| Formula | C21H24N4O2 |
| Molar mass | 364.449 g·mol−1 |
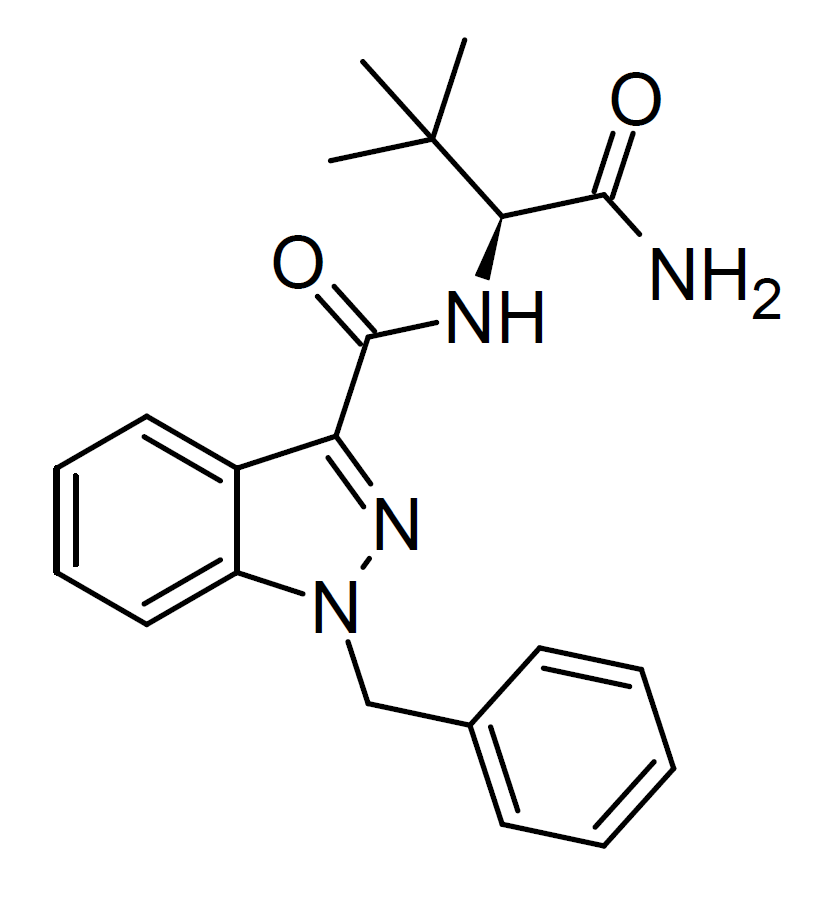
ADB-BUTINACA
The analog featuring a 1-butyl substitution on the indazole ring, as opposed to the 1-benzyl variant, was previously marketed as a designer drug under the designation ADB-BINACA. However, to eliminate potential confusion with the benzyl compound, it is now more commonly known as ADB-BUTINACA. This alternative compound exhibits a comparable potency as a CB1 agonist, boasting a remarkable binding affinity of 0.29nM for CB1 and 0.91nM for CB2, along with an EC50 of 6.36 nM for CB1.
FAQ
1. What is ADB-BINACA?
- ADB-BINACA is a synthetic cannabinoid designer drug. It has been used as an ingredient in some synthetic cannabis products.
2. How was ADB-BINACA developed?
- Pfizer initially developed ADB-BINACA as a potential analgesic. However, it later found its way into the realm of designer drugs.
3. What are the effects of ADB-BINACA?
- The effects of ADB-BINACA can vary widely and may include psychoactive properties similar to those of natural cannabinoids. However, it can also be associated with adverse effects, and its usage may be harmful.
4. Is ADB-BINACA legal?
- The legality of ADB-BINACA varies by country and jurisdiction. In many places, it is considered illegal due to its potential for misuse and harm.
5. What are the health risks associated with ADB-BINACA use?
- ADB-BINACA use is associated with various health risks, including potential addiction, adverse psychological effects, and physical harm. Long-term health consequences may be severe.
6. Can ADB-BINACA be detected in drug tests?
- Yes, ADB-BINACA can be detected in drug tests designed to identify synthetic cannabinoids. It’s important to note that these tests are becoming more sophisticated in detecting such substances.
7. What is the difference between ADB-BINACA and ADB-BUTINACA?
- ADB-BUTINACA is a related compound with a 1-butyl substitution on the indazole ring, in contrast to the 1-benzyl substitution in ADB-BINACA. Both are potent CB1 receptor agonists, but ADB-BUTINACA is designed to avoid confusion with the benzyl compound.
8. Is ADB-BINACA safe for recreational use?
- No, ADB-BINACA and similar designer drugs are not safe for recreational use. They can have unpredictable and potentially dangerous effects on the body and mind.
9. How can I seek help if I or someone I know is using ADB-BINACA?
- Suppose you or someone you know is using ADB-BINACA and needs assistance. In that case, it is crucial to seek help from a medical professional, addiction counselor, or a local substance abuse support group.
10. Where can I find more information about ADB-BINACA?
- For in-depth information, you can consult credible sources such as medical professionals, government health agencies, and addiction support organizations. Always ensure the information you access is up-to-date and from reliable sources.
References
- Qian Z, Hua Z, Liu C, and Jia W (2016) delved into four distinct varieties of cannabimimetic indazole and indole derivatives, namely, ADB-BINACA, AB-FUBICA, ADB-FUBICA, and AB-BICA, identifying them as novel psychoactive substances. This research can be found in “Forensic Toxicology” (Volume 34) with the article spanning pages 133 to 143. The study is accessible via DOI: 10.1007/s11419-015-0297-2, and it is also indexed under PMC 4705129 with PMID 26793280.
- “Indazole Derivatives” is documented in WO 2009106982, focusing on this intriguing category of compounds.
- Anvisa, on March 31, 2023, published “RDC Nº 784,” which pertains to the “Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial” (Collegiate Board Resolution No. 784 – Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control) in Brazilian Portuguese. This publication can be traced back to April 4, 2023, and it was archived from the original on August 3, 2023. The retrieval date was August 15, 2023.
- Kavanagh P, Pechnikov A, Nikolaev I, Dowling G, Kolosova M, and Grigoryev A, in July 2022, conducted research on the detection of ADB-BUTINACA metabolites in human urine, blood, kidney, and liver. This study is accessible through “Journal of Analytical Toxicology” (Volume 46, Issue 6) on pages 641 to 650, with a DOI of 10.1093/jat/bkab088 and PMID 34341821.
- In November 2021, Sia CH, Wang Z, Goh EM, Tan YL, Fong CY, Moy HY, and Chan EC explored urinary metabolite biomarkers for detecting synthetic cannabinoid ADB-BUTINACA abuse in “Clinical Chemistry” (Volume 67, Issue 11), spanning pages 1534 to 1544. The article is accessible through DOI: 10.1093/clinchem/hvab134 and PMID 34387654.
- Kronstrand R, Norman C, Vikingsson S, Biemans A, Valencia Crespo B, Edwards D, and others conducted a study in April 2022 on the metabolism of synthetic cannabinoids ADB-BUTINACA and ADB-4en-PINACA. The results and detection in forensic toxicology casework and infused papers seized in prisons are documented in “Drug Testing and Analysis” (Volume 14, Issue 4) on pages 634 to 652. The study can be accessed through DOI: 10.1002/dta.3203 and PMID 34811926, with S2CID 244490343.
- Wang Y, Pan Y, Yang H, Liu J, Wurita A, and Hasegawa K, in July 2022, quantified MDMB-4en-PINACA and ADB-BUTINACA in human hair by gas chromatography-tandem mass spectrometry. This research can be found in “Forensic Toxicology” (Volume 40, Issue 2) on pages 340 to 348, with DOI: 10.1007/s11419-022-00615-z and PMID 36454410, along with S2CID 247501663.
- King A, Hill SL, Pucci M, Bailey G, Keating L, Macfarlane R, and others explored clinical features associated with ADB-BUTINACA exposure in patients attending emergency departments in England in October 2022. This valuable data is documented in “Clinical Toxicology” (Volume 60, Issue 10) on pages 1094 to 1098, with a DOI of 10.1080/15563650.2022.2101469 and PMID 35943421, along with S2CID 251444625.
- For more information regarding ADB-BUTINACA, you can refer to the following link: ADB-BUTINACA Information.
- Cannaert A, Sparkes E, Pike E, Luo JL, Fang A, Kevin RC, and others engaged in the synthesis and in vitro assessment of cannabinoid receptor 1 activity for various synthetic cannabinoids, including ADB-BINACA. Their findings are available in “ACS Chemical Neuroscience” (Volume 11, Issue 24) on pages 4434 to 4446, with a DOI of 10.1021/acschemneuro.0c00644 and PMID 33253529, along with S2CID 227246346.
- Pike E, Grafinger KE, Cannaert A, Ametovski A, Luo JL, Sparkes E, and others systematically evaluated a panel of 30 synthetic cannabinoid receptor agonists structurally related to MMB-4en-PICA, MDMB-4en-PINACA, ADB-4en-PINACA, and MMB-4CN-BUTINACA. This comprehensive assessment included synthesis, analytical characterization, and binding affinity for human CB1 receptors. The research is segmented into two parts, with the first part focused on “Drug Testing and Analysis” (Volume 13, Issue 7) on pages 1383 to 1401, featuring a DOI of 10.1002/dta.3037 and PMID 33787091.