Beautiful Plants For Your Interior
Summary
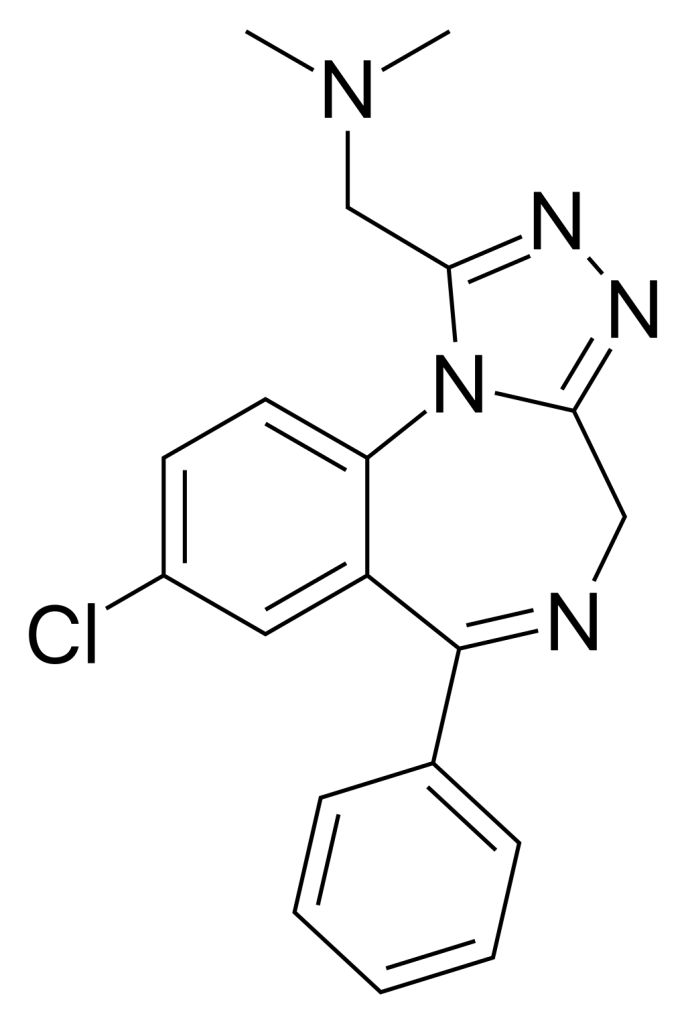
Adinazolam, marketed under the brand name Deracyn, is a tranquilizer belonging to the triazolobenzodiazepine (TBZD) class. This class combines traditional benzodiazepines (BZDs) with a triazole ring. Adinazolam is known for its anxiolytic, anticonvulsant, sedative, and antidepressant properties.
The drug’s development can be credited to Jackson B. Hester, who aimed to enhance the antidepressant effects of alprazolam, a medication he also played a role in developing. It’s worth noting that Adinazolam never received approval from the FDA and was never introduced to the public market. However, it has been illicitly sold as a designer drug.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 37115-32-5 |
|---|---|
| PubChem CID | 37632 |
| DrugBank | DB00546 |
| ChemSpider | 34519 |
| UNII | KN08449444 |
| KEGG | D02770 |
| ChEBI | CHEBI:251412 |
| ChEMBL | ChEMBL328250 |
| CompTox Dashboard (EPA) | DTXSID40190611 |
| Chemical and physical data | |
| Formula | C19H18ClN5 |
| Molar mass | 351.8 g·mol−1 |
Side effects
An overdose of adinazolam can result in various symptoms, including muscle weakness, ataxia, dysarthria, and, particularly in children, paradoxical excitement. More severe cases may lead to diminished reflexes, confusion, and even coma.
In a human study comparing the effects and abuse potential of adinazolam (at doses of 30 mg and 50 mg) with diazepam, lorazepam, and a placebo, it was observed that adinazolam induces significant mental and physical sedation, as well as heightened mental discomfort.
Pharmacodynamics and Pharmacokinetics
Adinazolam exerts its effects by binding to peripheral-type benzodiazepine receptors. These receptors interact allosterically with GABA receptors, acting as agonists to produce inhibitory effects.
Metabolism
The August 1984 issue of The Journal of Pharmacy and Pharmacology reported the presence of active metabolites of adinazolam. The primary metabolite is N-desmethyladinazolam (NDMAD), which exhibits roughly 25 times higher affinity for benzodiazepine receptors compared to its precursor. This difference in affinity accounts for the benzodiazepine-like effects following oral administration. Multiple N-dealkylations lead to the removal of the dimethylaminomethyl side chain, further explaining the difference in potency. Additionally, two other metabolites, alpha-hydroxyalprazolam and estazolam, were identified. In the same journal’s August 1986 issue, it was reported that proadifen inhibits the formation of N-desmethyladinazolam.
FAQ
- What is Adinazolam?
- Adinazolam is a tranquilizer classified under the triazolobenzodiazepine (TBZD) group. TBZDs are a subset of benzodiazepines (BZDs) with a triazole ring fused to their structure. Adinazolam exhibits anxiolytic (anxiety-reducing), anticonvulsant, sedative, and even antidepressant properties.
- Who developed Adinazolam?
- Adinazolam was developed by Jackson B. Hester, who aimed to enhance the antidepressant properties of alprazolam, another benzodiazepine he contributed to the development of.
- Was Adinazolam ever approved for use?
- Adinazolam never received approval from the U.S. Food and Drug Administration (FDA) and was never made available to the public market. However, it has been sold as a designer drug.
- What are the effects of an Adinazolam overdose?
- An overdose of Adinazolam can result in a range of symptoms, including muscle weakness, ataxia (lack of coordination), dysarthria (difficulty speaking), and paradoxical excitement, especially in children. In more severe cases, symptoms may include diminished reflexes, confusion, and even coma.
- How does Adinazolam compare to other benzodiazepines in terms of effects?
- A study comparing the subjective effects and abuse potential of Adinazolam with diazepam, lorazepam, and a placebo found that Adinazolam induces the most “mental and physical sedation” and the most significant “mental unpleasantness.”
- What is the primary active metabolite of Adinazolam?
- N-desmethyladinazolam (NDMAD) is the primary active metabolite of Adinazolam. It exhibits approximately 25 times higher affinity for benzodiazepine receptors compared to Adinazolam itself, contributing to its benzodiazepine-like effects after oral administration.
- Are there other notable metabolites of Adinazolam?
- Yes, apart from NDMAD, other metabolites include alpha-hydroxyalprazolam and estazolam.
- How does Adinazolam affect the central nervous system?
- Adinazolam acts by binding to peripheral-type benzodiazepine receptors, which then interact allosterically with GABA receptors, producing inhibitory effects in the central nervous system.
- Is Adinazolam available for medical use today?
- Adinazolam remains unapproved for medical use and is not a commonly prescribed medication.
- Why is Adinazolam considered a designer drug?
- Adinazolam has been marketed and sold as a designer drug, often without regulatory approval or oversight, making it available in specific illicit contexts.
References
- Patent for Adinazolam: A patent for adinazolam, titled “4,5-dihydro-4h-s-triazolo (4,3-a) (1,4) benzodiazepine – cns depressants, anti-convulsants, anti-aggressives and somatic reflex in,” was issued on January 21, 1977. The patent was assigned to Ciba-Geigy AG and Novartis AG.
- Hepatic Biotransformation of Adinazolam: A study conducted in March 1998 identified the enzymes responsible for the hepatic biotransformation of adinazolam and its active metabolite N-desmethyladinazolam in humans. This research provided insights into the metabolic pathways of adinazolam.
- Antidepressant Properties of Adinazolam: In June 1987, a double-blind study evaluated Adinazolam’s potential as a new antidepressant in outpatients with major depression. The study’s findings shed light on the medication’s effectiveness in treating depressive disorders.
- Pharmacological Profile of Adinazolam: In November 1983, research explored the pharmacological profile of adinazolam, highlighting its properties as a triazolobenzodiazepine. This study offered insights into the unique characteristics of adinazolam.
- Pharmaceutical Discoverers Award: Adinazolam was recognized with the Discovers Award in 2004. This award acknowledged significant contributions in the pharmaceutical field, including the development or research related to adinazolam.
- Designer Benzodiazepines Metabolism: In November 2016, a study characterized and examined the in vitro phase I microsomal metabolism of various designer benzodiazepines, including adinazolam. Understanding the metabolism of these substances is crucial for their regulation and detection.
- Adinazolam Information: You can find information about adinazolam on resources like DrugBank, providing valuable details about the drug’s properties and pharmacology.
- Abuse Potential Comparison: A study titled “The Abuse Potential of Adinazolam” compared the abuse potential of adinazolam with other benzodiazepines such as diazepam and lorazepam. This research aimed to evaluate the potential for misuse or dependence associated with adinazolam.
- Biological Activity and Metabolites: In August 1984, research determined the biological activity of adinazolam and its metabolites. Understanding the effects of these compounds helps in assessing their pharmacological relevance.
- Analytical Methods for Detection: Studies from the 1980s explored analytical methods for detecting adinazolam and its major metabolites in various biological samples. These methods are crucial for identifying and monitoring the presence of adinazolam in clinical and forensic settings.