Beautiful Plants For Your Interior
Summary
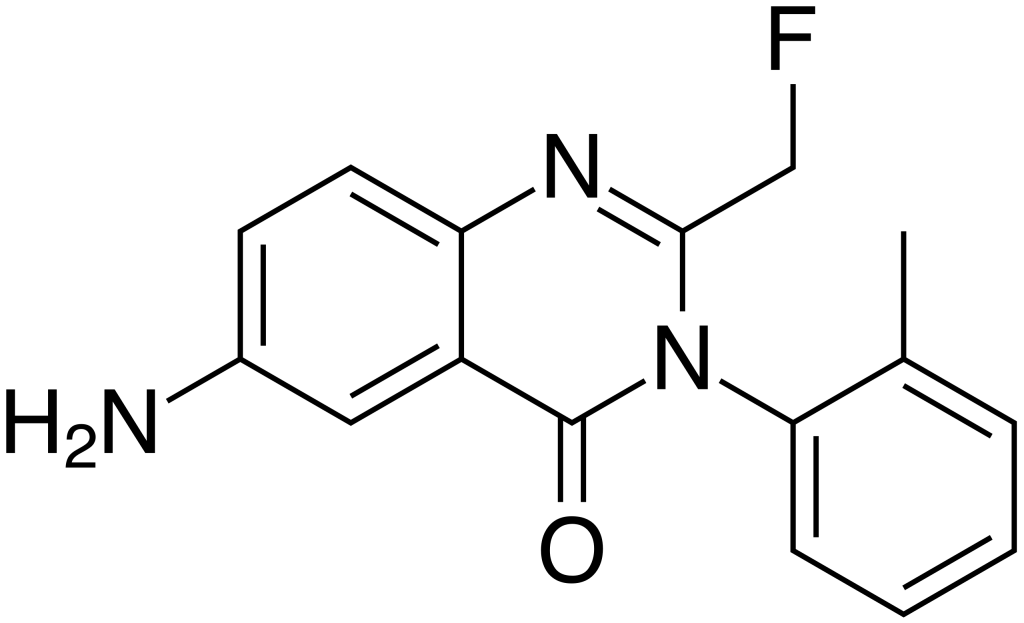
Afloqualone, also known as Arofuto, belongs to the quinazolinone family of GABAergic drugs. It is an analog of methaqualone that was developed in the 1970s by researchers at Tanabe Seiyaku. This compound exerts sedative and muscle-relaxant effects through its agonistic activity at the β subtype of the GABA receptor. Afloqualone has found some clinical applications, but it is associated with a side effect known as photosensitization, which can lead to skin issues like dermatitis.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 56287-74-2 |
|---|---|
| PubChem CID | 2040 |
| ChemSpider | 1960 |
| UNII | CO4U2C8ORZ |
| KEGG | D01638 |
| ChEMBL | ChEMBL2105918 |
| CompTox Dashboard (EPA) | DTXSID5022562 |
| Chemical and physical data | |
| Formula | C16H14FN3O |
| Molar mass | 283.3 g·mol−1 |
FAQ
- What is Afloqualone (Arofuto)?
- Afloqualone, commonly known as Arofuto, is a pharmaceutical compound belonging to the quinazolinone family of drugs. It is primarily used as a sedative and muscle relaxant.
- How does Afloqualone work?
- Afloqualone exerts its effects by acting as an agonist at the β subtype of the GABAa receptor in the central nervous system. This action leads to sedation and muscle relaxation.
- What are the clinical applications of Afloqualone?
- Afloqualone has found some clinical use in specific medical scenarios where sedation and muscle relaxation are required. Healthcare professionals may prescribe it for specific purposes.
- What is photosensitization, and how does it relate to Afloqualone?
- Photosensitization is a side effect associated with Afloqualone use. It means that individuals taking Afloqualone may become more sensitive to light, particularly sunlight. This heightened sensitivity can result in skin problems such as dermatitis.
- How can one minimize the risk of photosensitization while using Afloqualone?
- To reduce the risk of photosensitization, individuals using Afloqualone should take precautions when exposed to sunlight or other sources of ultraviolet (UV) light. This may include using sunscreen, wearing protective clothing, and avoiding excessive sun exposure.
- Is Afloqualone available over-the-counter or through prescription only?
- Afloqualone is typically available through prescription only. Its use should be under the guidance and supervision of a qualified healthcare professional.
- What are the potential side effects of Afloqualone besides photosensitization?
- Like many medications, Afloqualone may have other side effects, including drowsiness, dizziness, or digestive issues. It’s important to discuss any potential side effects with your healthcare provider.
- Can Afloqualone be used during pregnancy or while breastfeeding?
- The safety of using Afloqualone during pregnancy and breastfeeding has not been well established. It’s crucial to consult with a healthcare provider in such cases to assess potential risks and benefits.
- What precautions should one take while using Afloqualone?
- Individuals using Afloqualone should follow their healthcare provider’s instructions closely, be aware of potential side effects, and take necessary precautions against photosensitization. It’s also essential not to operate heavy machinery or drive when experiencing drowsiness or dizziness.
- Is Afloqualone known to interact with other medications or substances?
- Afloqualone may interact with certain medications or substances. It’s essential to inform your healthcare provider about all the medications, supplements, and substances you are using to prevent potential interactions.
References
- United States Patent US 3966731 describes the compound “2-Fluoromethyl-3-o-tolyl-6-amino-4(3H)-quinazolinone.” This patent was issued on June 29, 1976, and it is assigned to Tanabe Seiyaku Co Ltd.
- Inoue I, Oine T, Yamada Y, Tani J, Ishida R, and Ochiai T contributed to the development of this compound.
- Ochiai T and Ishida R conducted pharmacological studies on “6-amino-2-fluoromethyl-3-(O-tolyl)-4(3H)-quinazolinone,” also known as afloqualone, in June 1982. They investigated its effects on the spinal reflex potential and rigidity. This research was published in the Japanese Journal of Pharmacology, volume 32, issue 3, pages 427–438. The publication is identified by the DOI: 10.1254/jjp.32.427 and the PMID: 7109348.
- Ishikawa T, Kamide R, and Niimura M conducted research on afloqualone in June 1994, specifically on its role in inducing “Photoleukomelanodermatitis (Kobori).” This work was published in The Journal of Dermatology, volume 21, issue 6, on pages 430–433. The publication is identified by the DOI: 10.1111/j.1346-8138.1994.tb01768.x and the PMID: 8064007. Additionally, it is referenced with S2CID: 7486566.