Beautiful Plants For Your Interior
Summary
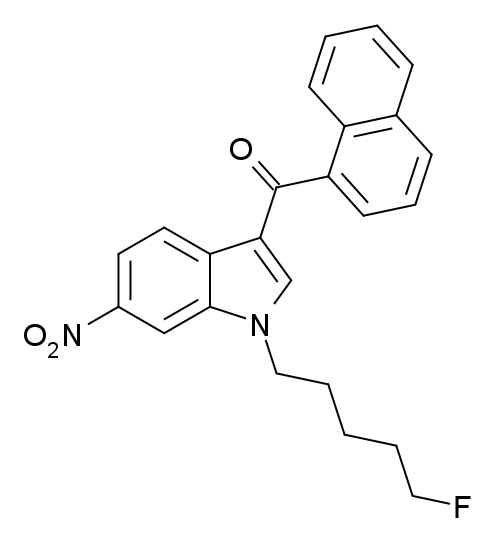
AM-1235, also known as 1-(5-fluorophenyl)-3-(naphthalene-1-oyl)-6-nitroindole, is a potent and moderately selective agonist targeting the cannabinoid receptor CB1.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 335161-27-8 |
|---|---|
| ChemSpider | 26633899 |
| UNII | 2HV9AH611M |
| CompTox Dashboard (EPA) | DTXSID10187159 |
| Chemical and physical data | |
| Formula | C24H21FN2O3 |
| Molar mass | 404.441 g·mol−1 |
Pharmacology
Pharmacodynamics
AM-1235 functions as a cannabinoid receptor agonist, with a Ki value of 1.5 nM at CB1 and 20.4 nM at CB2 receptors. A 6-nitro substitution on the indole ring reduces the affinity for both CB1 and CB2 when compared to the parent compound AM-2201. However, CB2 affinity is significantly reduced, resulting in approximately 13 times selectivity for CB1 over CB2. This is unlike other related compounds, such as AM-1221, where a 6-nitro substitution results in substantial selectivity for CB2.
Pharmacokinetics
This section needs additional citations for verification. Please help improve this article by adding citations to reliable sources in this section. Unsourced material may be challenged and removed. (July 2012) (Learn how and when to remove this template message).
The pharmacokinetic properties of AM-1235 are generally comparable to those of JWH-018.
AM-1235 undergoes metabolism that differs only slightly from that of JWH-018. N-dealkylation of AM-1235 produces fluoropentane, whereas JWH-018 generates pentane or plain alkanes. Fluoropentane might function as an alkylating agent or metabolize into toxic fluoroacetic acid. However, this is not the case, as fluoroalkanes do not typically act as alkylating agents under normal conditions. Uneven fluoroalkane chains usually metabolize into substantially less toxic fluoropropanoic acid.
Legal Status
In the United States, all CB1 receptor agonists belonging to the 3-(1-naphthyl)indole class, including AM-1235, are classified as Schedule I Controlled Substances.
FAQ
- What is AM-1235?
- AM-1235 is a synthetic drug known as a cannabinoid receptor agonist. It acts as an agonist for the cannabinoid receptor CB1 and has pharmacological effects on the endocannabinoid system.
- How does AM-1235 work?
- AM-1235 binds to the CB1 receptor in the endocannabinoid system, producing various effects. It primarily affects the CB1 receptor, but it also has some affinity for the CB2 receptor.
- What are the pharmacological properties of AM-1235?
- AM-1235 has a Ki (binding affinity) of 1.5 nM at the CB1 receptor and 20.4 nM at the CB2 receptor. This indicates that it has a higher affinity for the CB1 receptor, making it selective for CB1 over CB2. A 6-nitro substitution on the indole ring contributes to this selectivity.
- What are the pharmacokinetics of AM-1235?
- The pharmacokinetic properties of AM-1235 are similar to those of JWH-018. When metabolized, AM-1235 undergoes N-dealkylation, producing fluoropentane instead of pentane. Contrary to some speculation, fluoropentane is not an alkylating agent or metabolized into toxic fluoroacetic acid under normal conditions. Instead, it is likely metabolized into less harmful fluoropropanoic acid.
- Is AM-1235 legal?
- In the United States, AM-1235, along with all other CB1 receptor agonists of the 3-(1-naphthyl)indole class, is classified as a Schedule I Controlled Substance. This means it is illegal to possess, distribute, or use for recreational purposes.
- Is AM-1235 used for medical purposes?
- AM-1235 is not approved for medical use and is considered a designer drug. It is primarily used for research and recreational purposes, although its safety and legality for human consumption are subjects of concern and regulation.
- What are the potential risks associated with AM-1235 use?
- As a synthetic cannabinoid receptor agonist, AM-1235 can lead to a range of side effects and health risks. These may include altered perception, cognitive impairment, anxiety, paranoia, and potential unknown long-term health consequences.
- Is there ongoing research on AM-1235?
- Research on synthetic cannabinoids like AM-1235 is ongoing. However, their use and distribution are often illegal or restricted due to safety concerns and potential health risks.
- What should I do if I come into contact with AM-1235 or suspect its use?
- If you encounter AM-1235 or suspect its use, contact local law enforcement or relevant authorities, as it may be an illegal substance. The use of synthetic cannabinoids can be dangerous, and it’s essential to prioritize safety and legality.
References
- US Patent 7241799 was granted to Makriyannis A and Deng H for their work on “Cannabimimetic indole derivatives.” This patent was published on November 5, 2004, and officially issued on July 10, 2007.
- In the year 2000, Deng H completed their doctoral dissertation at the University of Connecticut. The title of the dissertation is “Design and synthesis of selective cannabinoid receptor ligands: Aminoalkylindole and other heterocyclic analogs.” The document can be found in ProQuest under the code 304624325.
- WO Patent 200128557 was granted to Makriyannis A and Deng H for their work on “Cannabimimetic indole derivatives.” This patent was published on April 26, 2001, and officially issued on June 7, 2001.
- In November 1956, Millington JE and Pattison FL published a paper titled “Toxic fluorine compounds: XII. Esters of ω-fluoroalcohols” in the Canadian Journal of Chemistry. This work can be found in Volume 34, Issue 11, on pages 1532–1541. The document is associated with the DOI: 10.1139/v56-200.
- In February 1957, Pattison FL, Howell WC, and Woolford RG published a paper titled “Toxic fluorine compounds: XIII. ω-Fluoroalkyl ethers” in the Canadian Journal of Chemistry. This work can be found in Volume 35, Issue 2, on pages 141–148. The document is associated with the DOI: 10.1139/v57-021.
- 21 U.S.C. § 812 outlines the Schedules of controlled substances in the United States.