Beautiful Plants For Your Interior
Summary
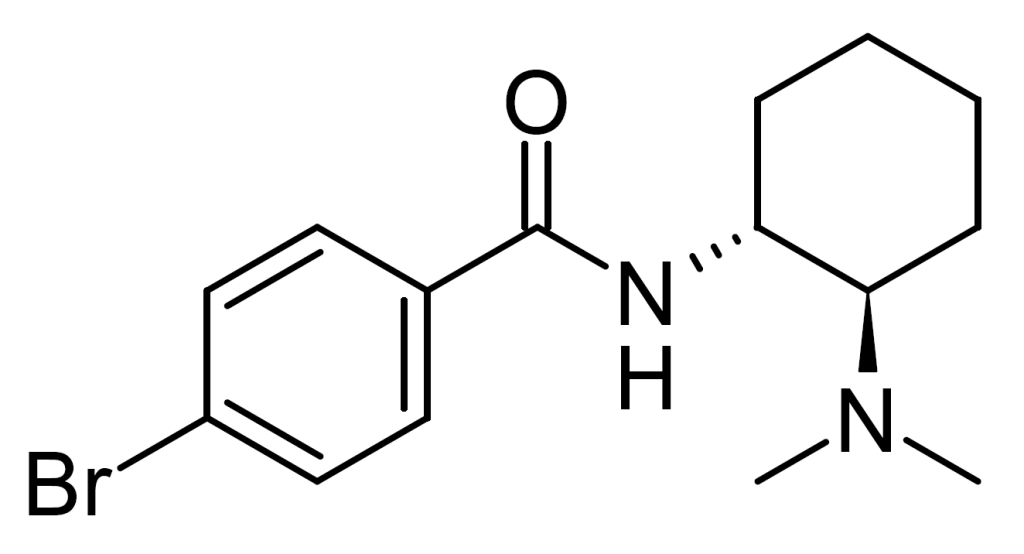
Bromadoline, also known as U-47931E, is a μ-opioid receptor-selective opioid analgesic created by Upjohn in the 1970s. It falls within the range of potency between codeine and morphine, with slight superiority to pentazocine. Bromadoline shares a chemical relationship with AH-7921 and U-47700.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 2418521-61-4 67579-24-2 (mixture of trans isomers) |
|---|---|
| PubChem CID | 6328449 |
| UNII | R8DWN01P1M |
| CompTox Dashboard (EPA) | DTXSID40217897 |
| Chemical and physical data | |
| Formula | C15H21BrN2O |
| Molar mass | 325.250 g·mol−1 |
FAQ
- What is Bromadoline?
- Bromadoline, also known as U-47931E, is an opioid analgesic drug developed by the Upjohn company in the 1970s. It selectively targets the μ-opioid receptor.
- How potent is Bromadoline?
- Bromadoline’s potency falls between that of codeine and morphine. It is slightly stronger than pentazocine.
- What are the similarities between Bromadoline and other opioids?
- Bromadoline is related to other opioids like AH-7921 and U-47700, sharing chemical similarities with these substances.
- Is Bromadoline commonly used for medical purposes?
- No, Bromadoline does not have typical medical applications. It was primarily developed for research purposes.
- Is Bromadoline legal?
- Legal statuses for substances can vary by country and region. It’s essential to check the local regulations in your area to determine its legality.
- What are the potential side effects of Bromadoline?
- Like other opioids, Bromadoline may cause side effects, including but not limited to sedation, constipation, respiratory depression, and addiction.
- Is Bromadoline safe for recreational use?
- No, the recreational use of Bromadoline or any opioid is considered risky. These substances can lead to severe health consequences, including overdose and death.
- Is Bromadoline associated with the opioid crisis?
- While Bromadoline is not as well-known as some opioids, it is related to substances that have been involved in the opioid crisis. The misuse of potent opioids has led to significant public health issues.
- Where can I find more information about Bromadoline?
- To stay updated on the latest information about Bromadoline, refer to reliable sources, such as scientific journals, government health agencies, and substance-specific resources.
- Is it legal to buy or sell Bromadoline online?
- The legal status of purchasing or selling Bromadoline online can vary widely based on your location. Be sure to understand and follow the laws and regulations applicable in your region.
References
- Unlisted Drugs Resource An invaluable resource for unlisted drugs can be found in “Unlisted Drugs,” a publication of the Chatham Pharmaceutical Section under the Special Libraries Association in 1982. This publication offers insights into various unlisted pharmaceutical compounds.
- Selective Opioid Receptor Research In March 1987, research by Hayes AG, Skingle M, and Tyers MB delved into the selectivity of opioid drugs by exploring the blocking effects of beta-funaltrexamine on their influence on urine output. This work contributed to a deeper understanding of the mechanisms of opioid receptor action.
- Patent US4098904 The Upjohn Company filed Patent US4098904 on July 4, 1978, which relates to analgesic N-(2-aminocycloaliphatic)benzamides. This patent is a notable milestone in the development of novel analgesic compounds.
- Differential Sensitivity of Antinociception Models In August 1987, Hayes AG, Sheehan MJ, and Tyers MB conducted research into the differing sensitivity of antinociception models in rats, mice, and guinea pigs when exposed to mu- and kappa-opioid receptor agonists. This study contributed to our understanding of the varying responses to opioid compounds across different animal models.
- Factors Affecting Binding of Trans-N-[2-(Methylamino)cyclohexyl]benzamides In December 1985, a study by Cheney BV, Szmuszkovicz J, Lahti RA, and Zichi DA explored the factors influencing the binding of trans-N-[2-(methylamino)cyclohexyl]benzamides at the primary morphine receptor. This research provided insights into the molecular interactions of this class of compounds at opioid receptors.