The market situation for 3-FEA, a prominent designer drug and research chemical, has been evolving in recent years. As with many research chemicals, 3-FEA has found its niche in the online marketplace. Sellers and vendors catering to the demand for novel compounds have made 3-FEA available for purchase on various online platforms.
This synthetic compound has piqued the interest of researchers and enthusiasts alike due to its potential effects and properties. As a result, it’s not uncommon to find 3-FEA listed for sale by reputable vendors who specialize in research chemicals. Buyers interested in acquiring 3-FEA can easily find it online through these vendors, which offer it for research purposes.
However, the market situation has its complexities. Regulatory authorities in various regions have implemented measures to control the sale and distribution of designer drugs, including 3-FEA. This has led to periodic changes in availability and legality, with some vendors discontinuing sales in response to evolving regulations.
Contents
- 1 Summary
- 2 Chemistry
- 3 Pharmacology
- 4 Subjective effects
- 5 Toxicity
- 6 Legal status
- 7 FAQ
- 7.1 1. What is 3-FEA?
- 7.2 2. Is 3-FEA legal?
- 7.3 3. What are the effects of 3-FEA?
- 7.4 4. Is 3-FEA safe to use?
- 7.5 5. What are the risks and side effects of 3-FEA?
- 7.6 6. Is 3-FEA addictive?
- 7.7 7. How should 3-FEA be dosed?
- 7.8 8. What is the duration of the 3-FEA effects?
- 7.9 9. Can 3-FEA be combined with other substances?
- 7.10 10. Is there any medical use for 3-FEA?
- 7.11 11. Where can I purchase 3-FEA?
- 7.12 12. What should I do if I experience adverse effects from 3-FEA?
- 8 References
Summary
3-Fluoroethamphetamine, often referred to as 3-FEA, represents a novel amphetamine compound featuring a ring substitution. When administered, it elicits a combination of entactogenic and stimulant effects. Structurally, 3-FEA shares a connection with a series of designer fluorinated amphetamines, including 2-FA, 2-FMA, 3-FA, 4-FMA, and 4-FA.
Similar to its precursor, 3-FA, the precise pharmacological, toxicological, and subjective impacts of 3-FEA in human subjects remain largely uncharted territory. Informal accounts have described 3-FEA as a moderately potent serotonin-focused triple monoamine releaser, resulting in a blend of entactogenic sensations and mild stimulation.
Despite its emergence, 3-FEA’s history within recreational human usage is notably brief, with no documented instances of street-level distribution. Instead, it has become accessible through online vendors operating in the grey market, offering it as a research chemical.
Given its robust psychostimulant properties, potential for habit formation, and uncertain toxicity profile, individuals considering the use of this substance are strongly advised to implement sound harm reduction practices for their safety and well-being.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 54982-43-3 |
|---|---|
| PubChem CID | 57458869 |
| ChemSpider | 27050449 |
| UNII | 7ZY9F4B6GT |
| Chemical and physical data | |
| Formula | C11H16FN |
| Molar mass | 181.254 g·mol−1 |
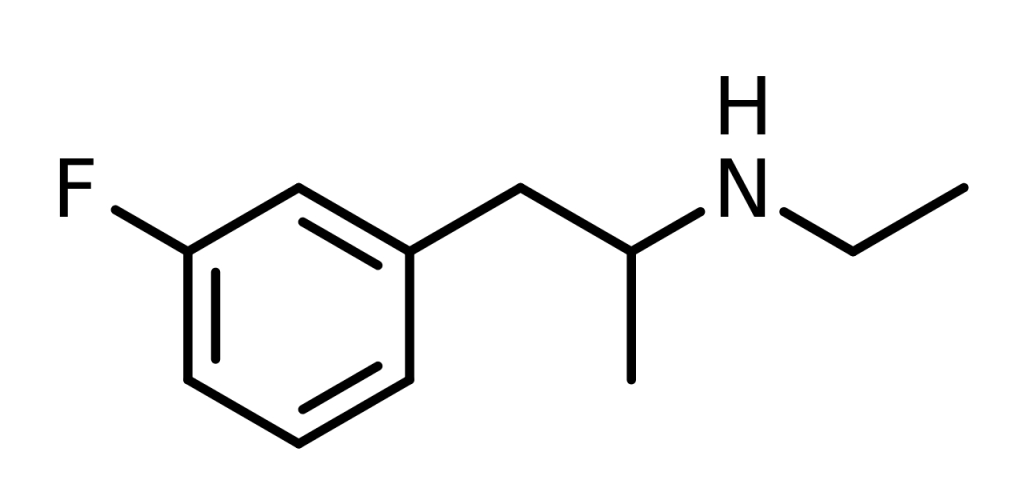
Chemistry
3-Fluoroethamphetamine, denoted as 3-FEA, is a synthetic compound belonging to the amphetamine chemical category. Amphetamines, as a chemical class, share a phenethylamine core structure, which encompasses a phenyl ring connected to an amino (NH2) group through an ethyl chain, bearing a methyl substitution at the Rα position. Essentially, amphetamines are phenethylamines that have undergone alpha methylation.
In specific terms, 3-FEA represents the fluorinated derivative of ethylamphetamine, also recognized as ethamphetamine. It further serves as an analog of fenfluramine, featuring the replacement of the 3-trifluoromethyl group with a 3-fluoro substituent.
Pharmacology
While 3-FEA hasn’t undergone extensive formal research akin to traditional amphetamines, it is currently postulated that, like other substituted amphetamines with analogous substitutions, its primary mode of action likely involves acting as a triple reuptake inhibitor and releaser of the monoamine neurotransmitters serotonin, dopamine, and norepinephrine.
Studies have demonstrated that in comparison to the unsubstituted ethylamphetamine, 3-fluoroethamphetamine exhibits reduced dopamine-releasing properties but enhanced release of both serotonin and norepinephrine. Intriguingly, it has displayed the most potent reinforcing effects in animal trials among a variety of 3-substituted amphetamine derivatives despite not being the most potent dopamine releaser.[3][2]
This suggests that 3-FEA effectively elevates the levels of the three major monoamine neurotransmitters—dopamine, norepinephrine, and serotonin—in the brain. It achieves this by functioning as a releasing agent for these neurotransmitters and by binding to and partially obstructing the transporter proteins responsible for clearing these molecules from the synaptic cleft after facilitating neural impulses. This blockade allows these molecules to accumulate in vital synaptic regions of the brain, resulting in a blend of relaxation, stimulation, disinhibition, and euphoria—a characteristic profile akin to entactogenic substituted amphetamines such as MDMA or 4-FA.

Subjective effects
Unlike its close analog, 3-FA, which has been reported as relatively functional and non-recreational, 3-FEA appears to yield effects more akin to another analog, 4-FA, known for its pronounced entactogenic effects. 3-FEA has also been noted to induce less stimulation compared to 4-FA, to the extent that some users describe it as primarily sedating. This effect profile positions 3-FEA as less suitable for functional use and better aligned with recreational purposes, similar to MDMA.
Please note that the following effects are based on anecdotal user reports and the Subjective Effect Index (SEI), which draws from the open research literature. They should be regarded with a degree of skepticism, as they may not occur predictably or consistently. Higher doses are more likely to encompass the full spectrum of effects, but they also carry a heightened risk of adverse outcomes, including addiction, severe injury, or even death ☠.
Physical:
- Sedation & Stimulation: 3-FEA is known to produce a paradoxical blend of soothing and stimulating effects. The sedative aspects are believed to result from its significant serotonin release, while stimulation becomes more prominent at dosages in the substantial range, particularly after the peak effects have subsided.
- Perception of Bodily Heaviness: Depending on the balance between stimulation and sedation, users may perceive themselves as either lighter or heavier.
- Spontaneous Bodily Sensations: Early reports describe a moderate to euphoric solid tingling sensation throughout the body, especially at higher doses. This sensation gradually intensifies upon onset and reaches its peak during the peak phase.
- Physical Euphoria
- Tactile Enhancement: Typically more noticeable at higher doses.
- Abnormal Heartbeat (citation needed)
- Increased Heart Rate (citation needed)
- Increased Blood Pressure (citation needed)
- Increased Perspiration
- Temperature Regulation Suppression (citation needed)
- Headaches
- Vasoconstriction (citation needed)
- Dehydration
- Dry Mouth (citation needed)
- Difficulty Urinating: 3-FEA may lead to temporary difficulty with urination, with a higher likelihood and increased severity at higher doses.
- Appetite Suppression: This effect can persist long after the main products have subsided (citation needed).
- Pupil Dilation: Often significant, even at lower ordinary doses. At standard doses, pupil dilation is comparable to that of a usual MDMA dosage and can persist after the main effects have worn off (citation needed). This effect is attributed to 3-FEA’s substantial serotonin release (source required).
- Teeth Grinding: Similar to MDMA, teeth grinding is more likely with higher or multiple doses.
- Diarrhea: Some users have reported a laxative effect (citation needed).
Cognitive:
- Anxiety Suppression or Anxiety: 3-FEA typically exhibits anxiety-suppressing effects associated with serotonin-releasing entactogens, although anxiety may arise toward the end of the substance’s duration, especially at higher doses.
- Disinhibition: Similar to MDMA.
- Empathy, Affection, and Sociability Enhancement: These effects are reportedly significant, occurring at standard and higher doses, akin to a typical MDMA experience (citation needed).
- Ego Inflation
- Thought Acceleration or Thought Deceleration: Thought acceleration is expected during the come-up and initial peak, followed by minor to significant thought deceleration during the latter half of the mountain, particularly at higher doses.
- Cognitive Euphoria
- Increased Music Appreciation: This effect is often prominent at all doses (citation needed).
- Increased Libido
- Immersion Enhancement
- Motivation Enhancement
- Compulsive Redosing: This tendency is most prevalent during or after the peak effects.
- Time Distortion: Users may perceive time as passing more rapidly than usual when sober.
- Wakefulness: Typically occurs following higher or multiple doses.
After:
Aftereffects during the offset of a stimulant experience are generally negative and uncomfortable compared to the peak effects. Commonly referred to as a “comedown,” this phase is a result of neurotransmitter depletion and may include:
- Anxiety
- Cognitive Fatigue
- Depression
- Irritability
- Motivation Suppression
- Pupil Dilation
- Thought Deceleration
- Wakefulness
Toxicity
The toxicity and long-term health consequences of recreational 3-FEA use remain primarily unstudied in a scientific context, and the precise toxic dose is yet unknown. This knowledge gap arises from 3-FEA’s brief history of human use, with its availability dating back only to mid-2016.
Initial anecdotal reports indicate that using this substance cautiously and sparingly at low to moderate doses appears to be devoid of adverse health effects. However, it is essential to exercise caution and employ proper harm-reduction practices when experimenting with 3-FEA.
Due to its serotonin-releasing entactogenic properties, there is a possibility that 3-FEA may exhibit significant affinity and activity at the 5-HT2B receptor. This similarity to 5-HT2B agonists like MDMA and fenfluramine suggests that long-term or heavy use may pose cardiotoxic risks.
Tolerance and Addiction Potential
Although the full extent is yet to be determined, chronic use of 3-FEA is likely to be regarded as moderately addictive, carrying a high potential for abuse and the potential to induce psychological dependence in certain users. Dependence or addiction may manifest as cravings and withdrawal symptoms upon discontinuation.
Tolerance to many of 3-FEA’s effects develops with prolonged and repeated use. This necessitates larger doses to achieve the same impact. After cessation, it typically takes 2 to 3 days for the tolerance to reduce by half and 3 to 7 days to return to baseline, assuming no further consumption. Cross-tolerance with other dopaminergic and serotonergic stimulants and entactogens is expected, meaning that the effects of all stimulants will be diminished (including atypical stimulants like MDMA or amphetamine due to their reliance on robust dopamine and norepinephrine stores to exert their full spectrum of effects).
Psychosis
The abuse of substances within the amphetamine class, particularly at high doses and over extended periods, can potentially lead to stimulant psychosis. This condition may manifest with symptoms such as paranoia, hallucinations, or delusions. Research suggests that a portion (approximately 5-15%) of users may not fully recover from amphetamine-induced psychosis. Antipsychotic medications have shown efficacy in treating acute amphetamine psychosis, which rarely arises from therapeutic use.
Dangerous Interactions
Caution should be exercised when combining 3-FEA with other substances, as dangerous interactions can occur. Some known interactions to be aware of include:
- Alcohol: Combining alcohol with stimulants can be risky, as it reduces alcohol’s sedative effects and may lead to excessive drinking, increasing the risk of harm to the liver and dehydration. It can also allow individuals to consume more alcohol than they would otherwise, potentially leading to dangerous levels of intoxication.
- GHB/GBL: Stimulants can increase respiration rate, allowing a higher dose of sedatives. If the stimulant’s effects wear off before the sedative’s effects, respiratory arrest may occur.
- Opioids: Like GHB/GBL, stimulants can increase respiration rate when combined with opioids. If the stimulant effects diminish before the opioids, respiratory arrest may result.
- Cocaine: Combining cocaine with amphetamine can lead to cardiac effects due to serotonin-related valvulopathy. It may also result in hypertension and an increased risk of syncope.
- Cannabis: The combination of stimulants and cannabis can heighten anxiety levels, increase the risk of thought loops, and induce paranoia.
- Caffeine: This combination is generally considered unnecessary and may place additional strain on the heart, potentially causing anxiety and physical discomfort.
- Tramadol: Both tramadol and stimulants elevate the risk of seizures.
- DXM: These substances both raise heart rate, and in extreme cases, panic attacks triggered by their combination can lead to more severe heart issues.
- Ketamine: Combining amphetamine and ketamine may result in psychoses resembling schizophrenia, albeit not necessarily more severe than those induced by each substance individually.
- PCP and Methoxetamine: These combinations increase the risk of tachycardia, hypertension, and manic states.
- Psychedelics (e.g., LSD, mescaline, psilocybin): Combining stimulants with psychedelics may raise the risk of anxiety, paranoia, and thought loops.
- 25x-NBOMe, 2C-T-x, 5-MeO-xxT, DOx, aMT, and MAOIs: These interactions may have a variety of effects, including hypertensive crises or thought disorders, depending on the specific substances involved.
Legal status
3-FEA currently occupies a legal gray area in many parts of the world. Its regulatory status is unclear, and it is not explicitly classified as illegal (“scheduled”) within any country’s legal framework. However, it’s essential to note that individuals could potentially face charges for its possession under specific circumstances, such as those related to analog laws or intent to sell or consume.
Here is the regulatory status of 3-FEA in various countries:
- Austria: 3-FEA is illegal to produce and sell under the Neue-Psychoaktive-Substanzen-Gesetz Österreich (NPSG).
- Canada: 3-FEA would likely be classified as Schedule I due to its analog status related to Amphetamine.
- Germany: 3-FEA is regulated under the New Psychoactive Substances Act (NpSG) as of November 26, 2016. Production, import with the intent to market, administration to others, and trading are punishable offenses. Possession is illegal but not penalized.
- New Zealand: 3-FEA is categorized as a Schedule 3 controlled substance in New Zealand, given its status as an amphetamine analog.
- Switzerland: 3-FEA may be considered a controlled substance as a defined derivative of α-methylphenethylamine under Verzeichnis E point 130. It is legal for scientific or industrial purposes.
- United Kingdom: 3-FEA is classified as a Class A drug due to the amphetamine analog clause within the Misuse of Drugs Act 1971.
- United States: 3-FEA could potentially be regarded as an analog of Amphetamine under the Federal Analogue Act, which might classify it as a Schedule II drug. The Federal Analogue Act, found in 21 U.S.C. § 813, is a section of the United States Controlled Substances Act. It treats any chemical considered “substantially similar” to an illegal drug in Schedule I or II as if it were also in those schedules. However, this applies only if the substance is intended for human consumption.
Individuals must stay informed about the legal status of 3-FEA in their respective regions, as these regulations can change over time. Additionally, it’s advisable to comply with local laws and regulations and to prioritize safety and responsible use when dealing with substances in this legal gray area.
FAQ
1. What is 3-FEA?
3-FEA, or 3-Fluoroethamphetamine, is a research chemical belonging to the substituted amphetamines class. It is structurally related to amphetamines and shares some similarities in effects.
2. Is 3-FEA legal?
The legal status of 3-FEA varies from country to country and region to region. It may be controlled or prohibited in some areas. Always check your local and national laws regarding the possession and use of research chemicals.
3. What are the effects of 3-FEA?
The results of 3-FEA are similar to other amphetamines and include increased energy, alertness, enhanced mood, and heightened sociability. Some users also report mild euphoria and empathy.
4. Is 3-FEA safe to use?
The safety of 3-FEA needs to be well-documented, as it is a relatively new research chemical. There are potential risks associated with its use, including cardiovascular issues, increased blood pressure, and possible long-term health effects. It should be used with caution and under the guidance of a qualified researcher.
5. What are the risks and side effects of 3-FEA?
Common side effects may include increased heart rate, elevated blood pressure, sweating, insomnia, anxiety, and jitteriness. Long-term or excessive use may lead to more severe health issues.
6. Is 3-FEA addictive?
As with many stimulants, 3-FEA has the potential for abuse and addiction, mainly if used regularly or in large doses. It’s essential to use it responsibly and be aware of the risk of developing a dependency.
7. How should 3-FEA be dosed?
Dosage recommendations for 3-FEA can vary widely depending on individual tolerance, body weight, and the purity of the substance. Start with a low dose and gradually titrate up if necessary. Always follow harm reduction practices and use accurate measuring equipment.
8. What is the duration of the 3-FEA effects?
The effects of 3-FEA typically last around 4 to 6 hours, but this can vary among users. After the effects wear off, users may experience a “comedown” period characterized by fatigue, irritability, and other negative feelings.
9. Can 3-FEA be combined with other substances?
Combining 3-FEA with other substances, especially other stimulants or drugs, can increase the risks and potential for adverse reactions. It’s essential to avoid polydrug use and consult with a knowledgeable professional if you have questions about combining substances.
10. Is there any medical use for 3-FEA?
3-FEA is not approved for medical use and is primarily used in scientific research. It has yet to undergo the rigorous testing and safety assessments required for pharmaceutical drugs.
11. Where can I purchase 3-FEA?
The sale and purchase of 3-FEA can be restricted or illegal in many places. It is essential to be cautious when sourcing research chemicals and to ensure you are adhering to all local laws and regulations.
12. What should I do if I experience adverse effects from 3-FEA?
If you or someone you know experiences adverse effects or a medical emergency related to 3-FEA use, seek immediate medical attention. It’s also crucial to be honest with healthcare professionals about substance use to receive appropriate care.
References
- Rösner, P., Quednow, B., Girreser, U., Junge, T. (March 2005). “Isomeric Fluoro-methoxy-phenylalkylamines: a new series of controlled-substance analogs (designer drugs).” Forensic Science International. 148 (2–3): 143–156. doi:10.1016/j.forsciint.2004.05.003. ISSN 0379-0738.
- Tessel, R. E., Rutledge, C. O. (May 1976). “Specificity of the release of biogenic amines from isolated rat brain tissue as a function of the meta substituent of N-ethylamphetamine derivatives.” The Journal of Pharmacology and Experimental Therapeutics. 197 (2): 253–262. ISSN 0022-3565.
- Tessel, R. E., Woods, J. H. (May 1978). “Substituted N-ethylamphetamine self-injection responding in the rhesus monkey: structure-activity relationships.” The Journal of Pharmacology and Experimental Therapeutics. 205 (2): 274–281. ISSN 0022-3565.
- Rothman, R. B., Baumann, M. H., Savage, J. E., Rauser, L., McBride, A., Hufeisen, S. J., Roth, B. L. (5 December 2000). “Evidence for Possible Involvement of 5-HT 2B Receptors in the Cardiac Valvulopathy Associated With Fenfluramine and Other Serotonergic Medications”. Circulation. 102 (23): 2836–2841. doi:10.1161/01.CIR.102.23.2836. ISSN 0009-7322.
- Shoptaw, S. J., Kao, U., Ling, W. (21 January 2009). Cochrane Drugs and Alcohol Group, ed. “Treatment for amphetamine psychosis.” Cochrane Database of Systematic Reviews. doi:10.1002/14651858.CD003026.pub3. ISSN 1465-1858.
- Hofmann, F. G. (1983). A handbook on drug and alcohol abuse: the biomedical aspects (2nd ed ed.). Oxford University Press. ISBN 9780195030563.
- [FDA Label] – http://www.accessdata.fda.gov/drugsatfda_docs/label/2013/021303s026lbl.pdf
- Greenwald, M. K., Lundahl, L. H., Steinmiller, C. L. (December 2010). “Sustained Release d-Amphetamine Reduces Cocaine but not ‘Speedball’-Seeking in Buprenorphine-Maintained Volunteers: A Test of Dual-Agonist Pharmacotherapy for Cocaine/Heroin Polydrug Abusers.” Neuropsychopharmacology. 35 (13): 2624–2637. doi:10.1038/npp.2010.175. ISSN 0893-133X.
- Siciliano, C. A., Saha, K., Calipari, E. S., Fordahl, S. C., Chen, R., Khoshbouei, H., Jones, S. R. (10 January 2018). “Amphetamine Reverses Escalated Cocaine Intake via Restoration of Dopamine Transporter Conformation.” The Journal of Neuroscience. 38 (2): 484–497. doi:10.1523/JNEUROSCI.2604-17.2017. ISSN 0270-6474.
- Krystal, J. H., Perry, E. B., Gueorguieva, R., Belger, A., Madonick, S. H., Abi-Dargham, A., Cooper, T. B., MacDougall, L., Abi-Saab, W., D’Souza, D. C. (1 September 2005). “Comparative and Interactive Human Psychopharmacologic Effects of Ketamine and Amphetamine: Implications for Glutamatergic and Dopaminergic Model Psychoses and Cognitive Function.” Archives of General Psychiatry. 62 (9): 985. doi:10.1001/archpsyc.62.9.985. ISSN 0003-990X.
- Branch, L. S. (2022), Consolidated federal laws of Canada, Controlled Drugs and Substances Act.
- “Anlage NpSG” (in German). Bundesministerium der Justiz und für Verbraucherschutz. Retrieved December 19, 2019.
- “Gesetz zur Bekämpfung der Verbreitung neuer psychoaktiver Stoffe” (PDF) (in German). Bundesanzeiger Verlag. Retrieved December 19, 2019.
- “§ 4 NpSG” (in German). Bundesministerium der Justiz und für Verbraucherschutz. Retrieved December 19, 2019.
- Misuse of Drugs Act 1975 No 116 (as of 01 July 2022), Public Act – New Zealand Legislation.
- “Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien” (in German). Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved January 1, 2020.
- Misuse of Drugs Act 1971.