Beautiful Plants For Your Interior
Where to Buy Escaline Online – Trusted Vendors and Sources
If you’re considering buying Escaline online, it’s crucial to choose a reliable and reputable vendor. Many online stores offer Escaline for sale, but purchasing from trusted sources ensures that you receive a legitimate and high-quality product. Look for vendors with positive customer reviews, secure payment options, and clear shipping policies. Whether you’re in the USA, Canada, or another country, make sure the vendor ships discreetly and complies with local regulations for research chemicals.
How to Order Escaline – A Step-by-Step Guide for Safe Purchases
Ordering Escaline online is simple, but it’s important to follow the right steps to ensure a safe purchase. First, select a reputable Escaline shop that offers secure payment methods and ships to your region. Make sure to verify the vendor’s reputation by reading reviews and checking their product descriptions. Once you’ve chosen the quantity, proceed to checkout and ensure that the vendor provides tracking information so you can monitor the shipment’s progress.
Escaline for Sale – Best Deals and Discounts
If you’re looking for Escaline for sale, you can find competitive pricing and special discounts from various trusted vendors. Many suppliers offer bulk discounts or promotional offers for first-time buyers, allowing you to get the best deal while ensuring the product’s quality. Compare prices from different vendors to find the best offer and check for discounts such as free shipping or limited-time promotions to maximize your savings.
Escaline USA – Where to Buy Escaline in the United States
For customers in the USA, buying Escaline online is simple, with several vendors offering shipping within the United States. However, it is important to verify the legal status of Escaline in your state, as research chemicals can be subject to varying regulations depending on location. Always choose a vendor who complies with US laws, offers secure payment options, and ensures discreet shipping to guarantee your order arrives safely and without complications.
Summary
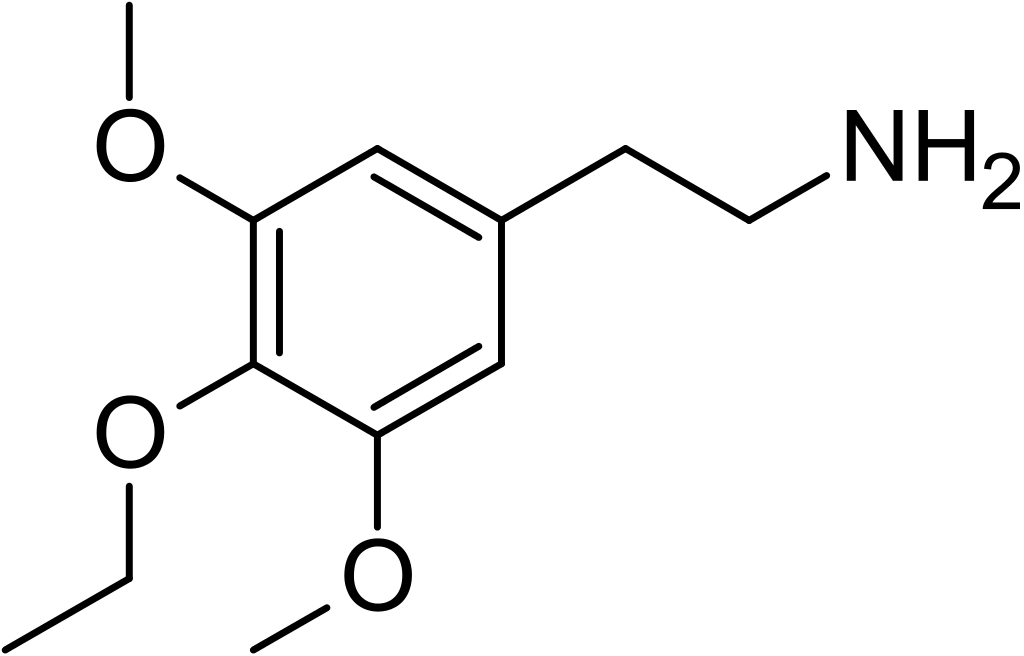
Escaline, scientifically known as 3,5-methoxy-4-ethoxyphenethylamine, is a notable psychedelic substance and entheogen within the phenethylamine class of compounds. Its presence in scientific literature dates back to 1954, when it was first synthesized and reported by Benington and colleagues. Subsequently, it garnered further attention in the laboratory of David E. Nichols, who conducted research on a range of mescaline analogs, including escaline, prescaling, and isoproscaline.
The effects of mescaline and related mescaline analogs in humans were comprehensively described by Alexander Shulgin, a pioneering figure in psychopharmacology. In his book PiHKAL (Phenethylamines I Have Known And Loved), Shulgin provides insights into mescaline, listing a recommended dosage range of 40 to 60 mg when taken orally. The duration of action is noted to be approximately 8 to 12 hours.
Escaline can be regarded as the phenethylamine counterpart to 3C-E and is recognized as the 4-ethoxy analog of mescaline.
| Identifiers | |
|---|---|
| CAS Number | 39201-82-6 |
| 3D model (JSmol) | Interactive imageInteractive image |
| ChEMBL | ChEMBL319415 |
| ChemSpider | 35053 |
| PubChemCID | 38240 |
| UNII | Q13F1C1N8I |
| CompTox Dashboard(EPA) | DTXSID60192455 |
Legal status
As of January 26, 2016, Escaline became prohibited in Sweden.
In the United States, Escaline is classified as a Schedule I controlled substance (DEA #7930) due to its status as a positional isomer of TMA (3,4,5-trimethoxyamphetamine).
FAQ
1. What is Escaline?
- Escaline is a psychedelic substance and entheogen that belongs to the phenethylamine class of compounds.
2. How was Escaline first discovered and researched?
- Escaline was first synthesized and reported in the scientific literature in 1954. It gained further attention through research conducted by David E. Nichols, who explored mescaline analogs, including escaline.
3. What are the effects and duration of Escaline?
- The effects of Escaline, as reported by Alexander Shulgin in his book PiHKAL, include psychedelic experiences. The duration of action is typically noted to be around 8 to 12 hours.
4. How is Escaline typically consumed?
- Escaline is usually taken orally, with a recommended dosage range of 40 to 60 mg, as listed by Alexander Shulgin.
5. Is Escaline legal?
- The legal status of Escaline varies by country and jurisdiction. It is considered illegal in some regions due to its classification as a controlled substance.
6. Are there any risks associated with Escaline use?
- Like other psychedelics, Escaline carries potential risks, including psychological distress and adverse effects. Responsible and informed use is essential.
7. Can Escaline be used for therapeutic purposes?
- There is limited research on the therapeutic potential of Escaline, and it is not approved for medical or therapeutic use. Other psychedelics, such as psilocybin and MDMA, have been more extensively studied for therapeutic applications.
8. Is Escaline addictive?
- The addictive potential of Escaline is not well understood, but it is generally considered to have a lower risk of physical dependence compared to substances like opioids or stimulants. Psychological dependence can still occur.
9. Where can I find more information about Escaline?
- Given the evolving legal and scientific landscape surrounding Escaline, it’s essential to consult reliable sources of scientific literature and seek advice from healthcare professionals. Staying informed and making well-informed decisions is crucial when considering the use of this substance.
References
- Alexander Shulgin’s PiHKAL (Phenethylamines I Have Known And Loved) includes a dedicated entry on Escaline, providing insights into its properties and effects.
- In November 2015, the Swedish public health agency, Folkhälsomyndigheten, made an announcement regarding the classification of substances, stating that 31 new substances could be classified as controlled substances or hazardous materials.
- For additional information regarding the classification and scheduling of substances, you can refer to the document available at this link: PDF.