Beautiful Plants For Your Interior
Summary
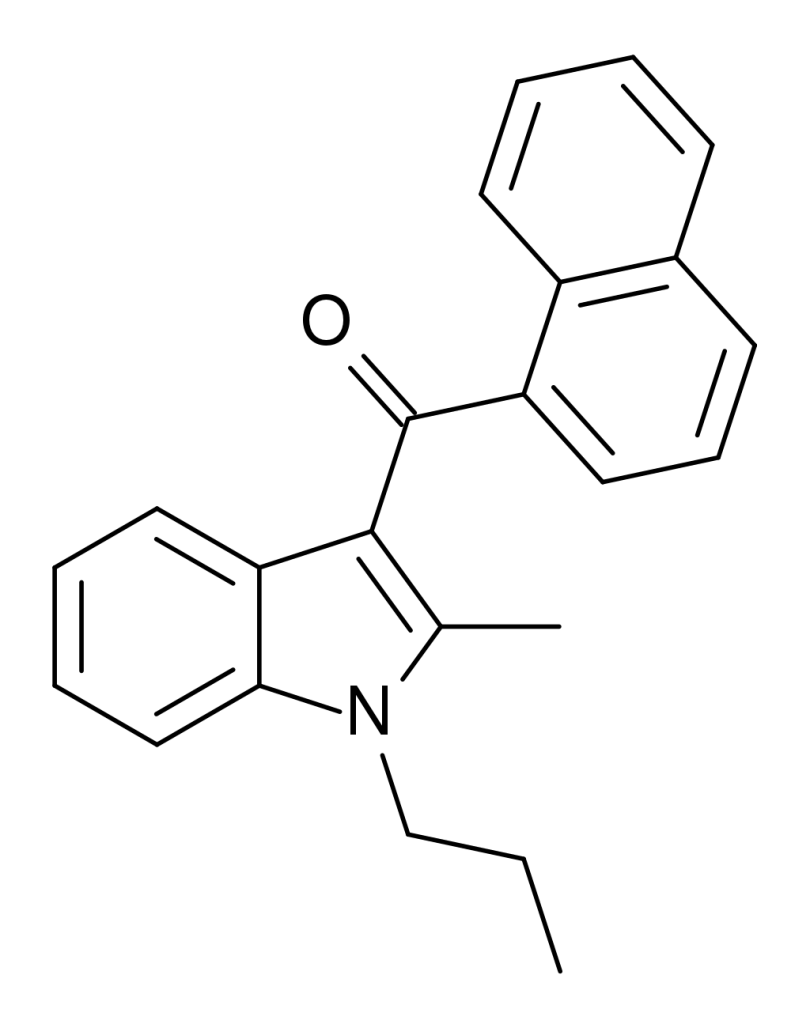
JWH-015, a member of the naphthoylindole family, functions as a cannabinoid agonist with subtype selectivity. Notably, it exhibits a high affinity for CB2 receptors, with a binding relationship of 13.8 nM, while its affinity for CB1 receptors is notably lower at 383 nM. This results in JWH-015 binding nearly 28 times more strongly to CB2 than CB1 receptors.
However, it’s important to note that JWH-015 does display some level of CB1 activity. In specific model systems, it can be remarkably potent and effective in activating CB1 receptors, making it less selective than newer drugs like JWH-133.
JWH-015 has demonstrated immunomodulatory properties, indicating its potential for influencing the immune system. CB2 receptor agonists like JWH-015 are being explored for their utility in addressing issues related to pain and inflammation.
This compound was initially discovered and named after the prominent researcher John W. Huffman.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 155471-08-2 |
|---|---|
| PubChem CID | 4273754 |
| IUPHAR/BPS | 5558 |
| ChemSpider | 3480676 |
| UNII | W4FL204T10 |
| CompTox Dashboard (EPA) | DTXSID60165902 |
| ECHA InfoCard | 100.161.912 |
| Chemical and physical data | |
| Formula | C23H21NO |
| Molar mass | 327.427 g·mol−1 |
Metabolism
JWH-015’s in vitro metabolism predominantly involves hydroxylation, N-dealkylation, and epoxidation of the naphthalene ring, following metabolic pathways akin to other aminoalkyl indole cannabinoids like WIN 55,212-2. It’s worth noting that the epoxidation of polycyclic aromatic hydrocarbons, as seen in compounds like benzo(a)pyrene, can yield carcinogenic metabolites. However, there’s no concrete evidence to suggest that JWH-015 or similar aminoalkylindole cannabinoids are carcinogenic in vivo. Notably, a study in the British Journal of Cancer indicates that JWH-015 might induce certain cancers to undergo apoptosis, potentially causing them to shrink.
Legal status
In the United States, substances that activate CB1 receptors and belong to the 3-(1-naphthyl)indole class, such as JWH-015, are categorized as Schedule I Controlled Substances.
Additionally, as of October 2015, JWH-015 is regulated as a controlled substance in China.
FAQ
- What is JWH-015?
- JWH-015 is a chemical compound from the naphthoylindole family, known for its properties as a cannabinoid agonist.
- How does JWH-015 affect cannabinoid receptors?
- JWH-015 primarily targets CB2 receptors with a high affinity (13.8 nM), but it also interacts with CB1 receptors, albeit with a lower affinity (383 nM).
- Is JWH-015 considered selective for CB2 receptors?
- While JWH-015 has a notable preference for CB2 receptors, it can still activate CB1 receptors effectively in some model systems. This makes it less selective compared to newer drugs like JWH-133.
- What are the potential medical uses of JWH-015?
- JWH-015 has shown immunomodulatory effects, and CB2 agonists like JWH-015 are being explored for their potential in addressing pain and inflammation issues.
- How is JWH-015 metabolized in the body?
- In vitro studies have revealed that JWH-015 is metabolized primarily through hydroxylation, N-dealkylation, and epoxidation of the naphthalene ring.
- Is JWH-015 carcinogenic?
- While the epoxidation of certain compounds can produce carcinogenic metabolites, there is currently no evidence to suggest that JWH-015 or similar aminoalkyl indole cannabinoids are carcinogenic in living organisms.
- Are there any studies on the effects of JWH-015 on cancer?
- Research published in the British Journal of Cancer suggests that JWH-015 may play a role in signalling certain cancers to undergo apoptosis, potentially causing them to shrink.
- What is the legal status of JWH-015?
- In the United States, substances like JWH-015, which activate CB1 receptors and belong to the 3-(1-naphthyl)indole class, are classified as Schedule I Controlled Substances. Additionally, as of October 2015, JWH-015 is a controlled substance in China.
- Who is John W. Huffman, and why is JWH-015 named after him?
- John W. Huffman is a notable researcher in the field of synthetic cannabinoids. JWH-015, along with many other JWH compounds, is named after him as he contributed significantly to the development and understanding of these compounds.
- Is JWH-015 approved for medical or recreational use?
- JWH-015 and similar compounds are not approved for medical or recreational use and may have unpredictable effects on health. It’s essential to be aware of local regulations and adhere to them.
References
- Aung MM, Griffin G, Huffman JW, Wu M, Keel C, Yang B, Showalter VM, Abood ME, Martin BR (August 2000). “Influence of the N-1 alkyl chain length of cannabimimetic indoles upon CB1 and CB2 receptor binding”. This study delves into the influence of alkyl chain length on the binding affinity of cannabimimetic indoles to CB1 and CB2 receptors, providing valuable insights into the structure-activity relationships of these compounds.
- Murataeva N, Mackie K, Straiker A (November 2012). “The CB2-preferring agonist JWH015 also potently and efficaciously activates CB1 in autaptic hippocampal neurons”. Explore how JWH015, known for its CB2 preference, can remarkably activate CB1 receptors in specific neurons, shedding light on its versatile pharmacological effects.
- Marriott KS, Huffman JW (2008). “Recent advances in the development of selective ligands for the cannabinoid CB(2) receptor”. Learn about recent advancements in the development of selective ligands targeting the CB2 receptor, an area of great interest for potential therapeutic applications.
- Ghosh S, Preet A, Groopman JE, Ganju RK (July 2006). “Cannabinoid receptor CB2 modulates the CXCL12/CXCR4-mediated chemotaxis of T lymphocytes”. Discover how the CB2 cannabinoid receptor influences the chemotaxis of T lymphocytes, offering insights into the modulation of immune responses.
- Montecucco F, Burger F, Mach F, Steffens S (March 2008). “CB2 cannabinoid receptor agonist JWH-015 modulates human monocyte migration through defined intracellular signaling pathways”. Explore the impact of the CB2 cannabinoid receptor agonist JWH-015 on human monocyte migration and the signaling pathways involved, potentially relevant to immune and inflammatory responses.
- Balter MB, Uhlenhuth EH (1992). “Prescribing and use of benzodiazepines: an epidemiologic perspective”. This study provides an epidemiologic perspective on the prescribing and use of benzodiazepines, offering insights into their medical and societal implications.
- Romero-Sandoval A, Eisenach JC (April 2007). “Spinal cannabinoid receptor type 2 activation reduces hypersensitivity and spinal cord glial activation after paw incision”. Learn about the effects of spinal cannabinoid receptor type 2 activation on reducing hypersensitivity and spinal cord glial activation, which may have implications for pain management.
- Zhang Q, Ma P, Cole RB, Wang G (Nov 2006). “Identification of in vitro metabolites of JWH-015, an aminoalkylindole agonist for the peripheral cannabinoid receptor (CB2) by HPLC-MS/MS”. Discover the in vitro metabolites of JWH-015, shedding light on its metabolic pathways and potential breakdown products.
- Zhang Q, Ma P, Iszard M, Cole RB, Wang W, Wang G (October 2002). “In vitro metabolism of R(+)-[2,3-dihydro-5-methyl-3-[(morpholinyl)methyl]pyrrolo [1,2,3-de]1,4-benzoxazinyl]-(1-naphthalenyl) methanone mesylate, a cannabinoid receptor agonist”. Explore the in vitro metabolism of R(+)-[2,3-dihydro-5-methyl-3-[(morpholinyl)methyl]pyrrolo [1,2,3-de]1,4-benzoxazinyl]-(1-naphthalenyl) methanone mesylate, a compound with potential cannabinoid receptor agonist activity.
- N Olea-Herrero; D Vara; S Malagarie-Cazenave; I Díaz-Laviada (18 August 2009). “Inhibition of human tumor prostate PC-3 cell growth by cannabinoids R(+)-Methanandamide and JWH-015: Involvement of CB2”. This study explores the inhibition of human tumor prostate PC-3 cell growth by cannabinoids R(+)-Methanandamide and JWH-015, with a focus on their involvement with CB2 receptors, potentially offering insights into cancer treatment.