Beautiful Plants For Your Interior
Summary
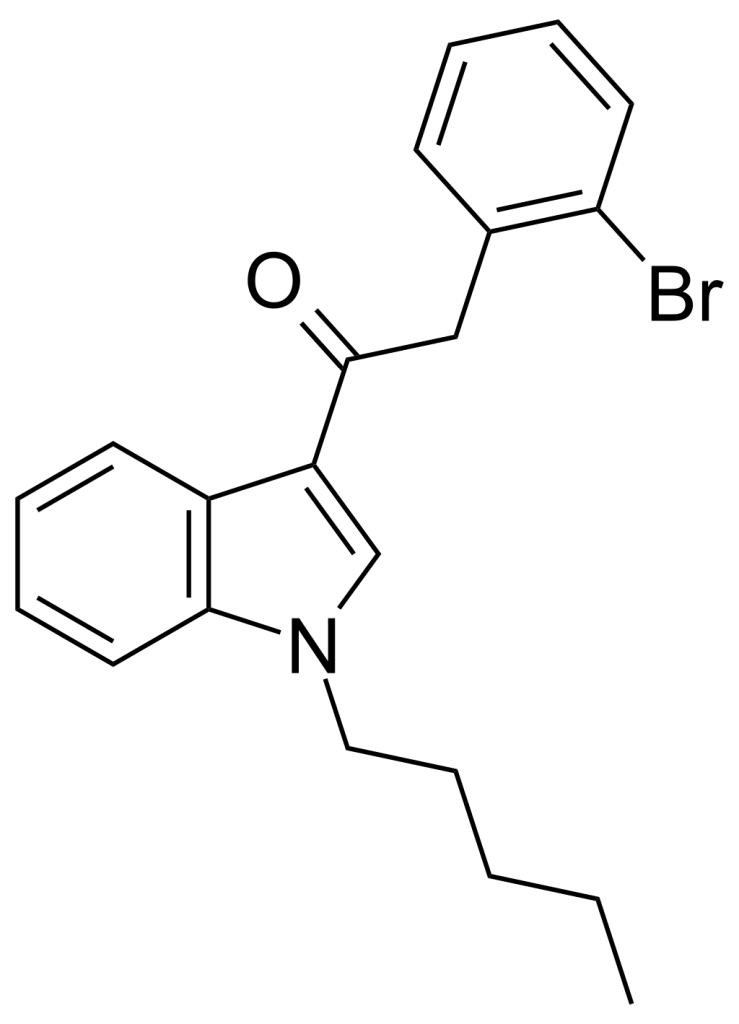
JWH-249, also known as 1-pentyl-3-(2-bromophenylacetyl)indole, is a synthetic cannabinoid belonging to the phenylacetylindole family. It operates as a cannabinoid agonist with approximately 2.4 times higher selectivity for CB1 receptors, having a Ki of roughly 8.4 ± 1.8 nM, compared to CB2 receptors, where its Ki is around 20 ± 2 nM. Much like its counterparts, including the 2′-methoxy compound JWH-250, the 2′-chloro compound JWH-203, and the 2′-methyl compound JWH-251, JWH-249 incorporates a phenyl acetyl group in place of the naphthyl ring typically found in most aminoalkyl indole cannabinoid compounds.
It is important to note that in the United States, CB1 receptor agonists falling under the 3-phenylacetylindole class, like cannabipiperidiethanone, are categorized as Schedule I Controlled Substances.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 864445-60-3 |
|---|---|
| PubChem CID | 44397335 |
| ChemSpider | 23253217 |
| UNII | H988KQV4U9 |
| ChEMBL | ChEMBL363471 |
| CompTox Dashboard (EPA) | DTXSID30658735 |
| Chemical and physical data | |
| Formula | C21H22BrNO |
| Molar mass | 384.317 g·mol−1 |
FAQ
1. What is JWH-249?
JWH-249 is a synthetic cannabinoid from the phenylacetylindole family, known for its cannabinoid agonist properties. It interacts with cannabinoid receptors in the body.
2. How does JWH-249 work?
JWH-249 functions as a cannabinoid agonist, binding to and activating cannabinoid receptors, particularly CB1. It exhibits a higher selectivity for CB1 compared to CB2 receptors.
3. What is its selectivity for CB1 and CB2 receptors?
JWH-249 demonstrates approximately 2.4 times greater selectivity for CB1 receptors, with a Ki (binding affinity) of roughly 8.4 ± 1.8 nM. At CB2 receptors, its Ki is about 20 ± 2 nM.
4. Are there similar compounds to JWH-249?
Yes, JWH-249 belongs to the same family as other synthetic cannabinoids, such as JWH-250, JWH-203, and JWH-251. These compounds share certain structural similarities.
5. How does JWH-249 differ from other synthetic cannabinoids?
JWH-249 distinguishes itself by the presence of a phenyl acetyl group in its chemical structure instead of the naphthyl ring typically found in most aminoalkyl indole cannabinoid compounds.
6. Is JWH-249 legal in the United States?
No, JWH-249 and other CB1 receptor agonists of the 3-phenylacetylindole class are classified as Schedule I Controlled Substances in the United States, making their manufacture, possession, or distribution illegal.
7. What are the potential effects and risks associated with JWH-249 use?
The effects and risks of using JWH-249 may vary, but like other synthetic cannabinoids, it can lead to altered perception, relaxation, and anxiety. Adverse effects can also occur, and the safety of its use is not well-established.
8. Is JWH-249 used for medical purposes?
JWH-249 is not approved for any medical use and is primarily associated with recreational or illicit use.
9. Where can I find more information about JWH-249?
You can explore scientific literature, drug regulation sources, and reputable educational websites for more information about JWH-249. Always ensure that your sources are credible and reliable when seeking information about such substances.
References
- In a groundbreaking study published in September 2005, Huffman JW and his colleagues unveiled a new class of compounds with remarkable cannabimimetic properties. These compounds, known as “1-Pentyl-3-phenylacetylindoles,” opened up exciting possibilities in the field of cannabinoid research. The findings were presented in the Bioorganic & Medicinal Chemistry Letters, Volume 15, Issue 18, Pages 4110–4113. [DOI: 10.1016/j.bmcl.2005.06.008, PMID: 16005223]
- Fast forward to April 2008 when Manera C, Tuccinardi T, and Martinelli A delved deeper into the world of cannabinoids and their ligands. Their comprehensive review shed light on “Indoles and Related Compounds as Cannabinoid Ligands,” providing essential insights into the interaction of these compounds with the endocannabinoid system. The mini-review was published in Mini Reviews in Medicinal Chemistry, Volume 8, Issue 4, Pages 370–387. [DOI: 10.2174/138955708783955935, PMID: 18473928]
- The legal framework that governs controlled substances in the United States can be found in 21 U.S.C. § 812. This regulation outlines the schedules of controlled substances, including those with potential medicinal uses and those deemed illicit due to their potential for abuse. Understanding this legal context is crucial in the study of compounds like cannabimimetic indoles.