Beautiful Plants For Your Interior
Summary
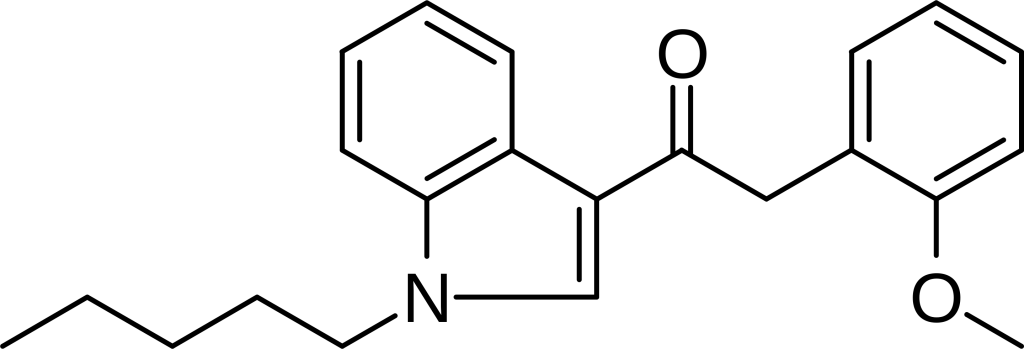
JWH-250, scientifically known as 1-pentyl-3-(2-methoxyphenylacetyl)indole, is a member of the phenylacetylindole family recognized for its analgesic properties. This chemical compound operates as a cannabinoid agonist, engaging with CB1 and CB2 receptors, featuring a Ki of approximately 11 nM at CB1 and 33 nM at CB2. It’s noteworthy that JWH-250 distinguishes itself from many older JWH series compounds by not incorporating a naphthalene ring. Instead, it occupies this position with a 2′-methoxy-phenylacetyl group, categorizing JWH-250 as a representative of a novel class of cannabinoid ligands. Furthermore, there are other active 2-substituted analogs, such as the methyl, chloro, and bromo compounds, which exhibit somewhat higher potency.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 864445-43-2 |
|---|---|
| PubChem CID | 44397540 |
| ChemSpider | 23256117 |
| UNII | IP9911R8A0 |
| ChEMBL | ChEMBL188031 |
| CompTox Dashboard (EPA) | DTXSID40235556 |
| Chemical and physical data | |
| Formula | C22H25NO2 |
| Molar mass | 335.447 g·mol−1 |
History
JWH-250, bearing the namesake of researcher John W. Huffman, was synthesized as part of his exploration into the structure and function of the mammalian endocannabinoid system. Its discovery, alongside several other compounds, aimed to further our understanding of this intricate system.
In May 2009, the German Federal Criminal Police identified JWH-250 in samples, which had been incorporated into new-generation “herbal smoking blends.” This occurred following the ban on the original ingredients, including (C8)-CP 47,497 and JWH-018.
Legal Status
Australia: JWH-250 is classified as a Schedule 9 prohibited substance in Australia as of October 2015. This designation indicates its potential for abuse or misuse, and its manufacture, possession, sale, or use is generally prohibited by law. Exceptions are made for medical or scientific research, as well as analytical, teaching, or training purposes, subject to approval by Commonwealth and State or Territory Health Authorities.
United States: In the United States, substances like JWH-250, belonging to the CB1 receptor agonists of the 3-phenylacetylindole class, are classified as Schedule I Controlled Substances. This categorization implies their legal prohibition, as substances in this schedule are deemed to have a high potential for abuse and no recognized medical use.
FAQ
1. What is JWH-250?
JWH-250 is a synthetic compound from the phenylacetylindole family. It acts as a cannabinoid agonist, interacting with cannabinoid receptors in the body.
2. Who discovered JWH-250, and why was it created?
JWH-250 was developed by researcher John W. Huffman to study the structure and function of the mammalian endocannabinoid system. It was part of a series of compounds created for this research.
3. When was JWH-250 first identified in “herbal smoking blends”?
Samples of JWH-250 were initially detected in May 2009 by the German Federal Criminal Police. It was found as an ingredient in new-generation “herbal smoking blends” that emerged following the prohibition of specific original components.
4. How does JWH-250 interact with the endocannabinoid system?
JWH-250 acts as a cannabinoid agonist, binding to CB1 and CB2 receptors in the endocannabinoid system.
5. What is the legal status of JWH-250 in Australia?
In Australia, JWH-250 is classified as a Schedule 9 prohibited substance under the Poisons Standard. This designation restricts its manufacture, possession, sale, or use, with exceptions for medical or scientific research and specific educational purposes, with proper approvals.
6. What is the legal status of JWH-250 in the United States?
In the United States, JWH-250 falls under Schedule I Controlled Substances. This classification means it is generally illegal, as substances in this schedule are considered to have a high potential for abuse and no recognized medical use.
7. Are there any similar compounds to JWH-250?
Yes, JWH-250 is part of the phenylacetylindole family, and there are other related compounds within this family, each with unique properties and interactions with cannabinoid receptors.
8. What are the potential effects and risks associated with JWH-250 use?
Effects and risks can vary, but like other synthetic cannabinoids, JWH-250 may lead to altered perception, relaxation, and anxiety. Adverse effects can also occur, and the safety of its use is not well-established.
9. Is JWH-250 used for medical purposes?
JWH-250 is not approved for any medical use and is primarily associated with recreational or illicit use.
10. Where can I find more information about JWH-250?
For more information about JWH-250, you can explore scientific literature, drug regulation sources, and reputable educational websites. Always ensure that your sources are credible and reliable when seeking information about such substances.
References
- Anvisa, in a resolution dated July 24, 2023, issued Collegiate Board Resolution No. 804, which delineates the Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control. This document, published in Brazilian Portuguese, is a crucial reference for regulatory control of certain substances, ensuring public safety and health. [Source: Diário Oficial da União, Archived from the original on August 27, 2023]
- In the realm of legal articles, the Latvian jurisdiction presents a comprehensive perspective on matters of legality and regulation. The Latvian legal system encompasses various aspects of law, including the control of substances and the classification of certain compounds.
- In a significant scientific development, the year 2005 witnessed a breakthrough. Huffman JW and his team introduced a new class of compounds in their publication titled “1-Pentyl-3-phenylacetylindoles.” These compounds, with cannabimimetic properties, opened doors to intriguing research in the field. The findings were published in the Bioorganic & Medicinal Chemistry Letters, Volume 15, Issue 18, Pages 4110–4113. [DOI: 10.1016/j.bmcl.2005.06.008, PMID: 16005223]
- Furthering our understanding of cannabinoids, in April 2008, Manera C, Tuccinardi T, and Martinelli A delved into “Indoles and Related Compounds as Cannabinoid Ligands.” This mini-review provided essential insights into the interaction of these compounds with the endocannabinoid system. The work was featured in Mini Reviews in Medicinal Chemistry, Volume 8, Issue 4, Pages 370–387. [DOI: 10.2174/138955708783955935, PMID: 18473928]
- “The Cannabinoid Receptors,” an edited work by Patricia H. Reggio, stands as a significant resource in the exploration of cannabinoid science. This book, bearing ISBN 978-1-58829-712-9, offers a comprehensive overview of cannabinoid receptors, their function, and the compounds that interact with them.
- The European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) delved into “Understanding the ‘Spice’ Phenomenon” in November 2009. This research and its findings offered valuable insights into the dynamics of synthetic cannabinoids, their use, and their potential impact. [Source: EMCDDA, Lisbon, November 2009, Archived from the original (PDF) on April 8, 2010]
- Arntson A, Ofsa B, Lancaster D, Simon JR, McMullin M, and Logan B contributed to the field of drug testing with their work on the validation of a novel immunoassay. This assay, designed for the detection of synthetic cannabinoids and their metabolites in urine specimens, presented a valuable tool for ensuring safety and compliance. The study was published in the Journal of Analytical Toxicology, Volume 37, Issue 5, Pages 284–290. [DOI: 10.1093/jat/bkt024, PMID: 23625703]
- The Poisons Standard for October 2015 is a crucial document outlining the regulation of substances in Australia. This standard, available for reference, plays a pivotal role in controlling the use and distribution of substances with the potential for abuse, safeguarding public health and well-being. [Source: Comlaw, Poisons Standard October 2015]
- The legal framework that governs controlled substances in the United States can be found in 21 U.S.C. § 812. This regulation outlines the schedules of controlled substances, including those with potential medicinal uses and those deemed illicit due to their potential for abuse. Understanding this legal context is crucial in the study of various compounds, their legality, and their impact on society.