Beautiful Plants For Your Interior
Where to Buy RCS-8 Online
For those looking to buy RCS-8 online, it’s important to purchase it from reliable research chemical vendors. Trusted suppliers offer RCS-8 for sale in the USA, Canada, and internationally. Before placing an order, ensure the vendor provides clear information about the product’s quality, purity, and shipping details. It’s essential to verify that the vendor adheres to safety and legal standards to guarantee that you’re purchasing a legitimate and safe research chemical. Many online shops provide RCS-8 with secure payment options and discreet shipping, making it easier to get the chemical you need for your studies.
RCS-8 for Sale in the USA and Canada
Researchers in the USA and Canada can easily find RCS-8 for sale from various trusted vendors. When purchasing RCS-8 in these countries, it’s crucial to check the legality of such purchases in your area, as research chemicals can be regulated differently depending on the region. Many vendors offer RCS-8 for sale with fast shipping to both the USA and Canada, allowing you to obtain the chemical you need for your research quickly and securely. Be sure to check for customer reviews and vendor reputation before making your purchase to ensure you’re buying a quality product.
How to Order RCS-8 Online Safely
Ordering RCS-8 online can be done securely by choosing trusted and verified vendors. It’s important to look for vendors that specialize in research chemicals and offer transparent product information, such as certificates of analysis, to guarantee the purity of the compound. Before finalizing your purchase, check the vendor’s reputation by reading customer feedback, ensuring their business practices align with your expectations for quality and security. Secure payment methods and discreet shipping are also crucial factors when ordering RCS-8, protecting both your financial and personal information.
Summary
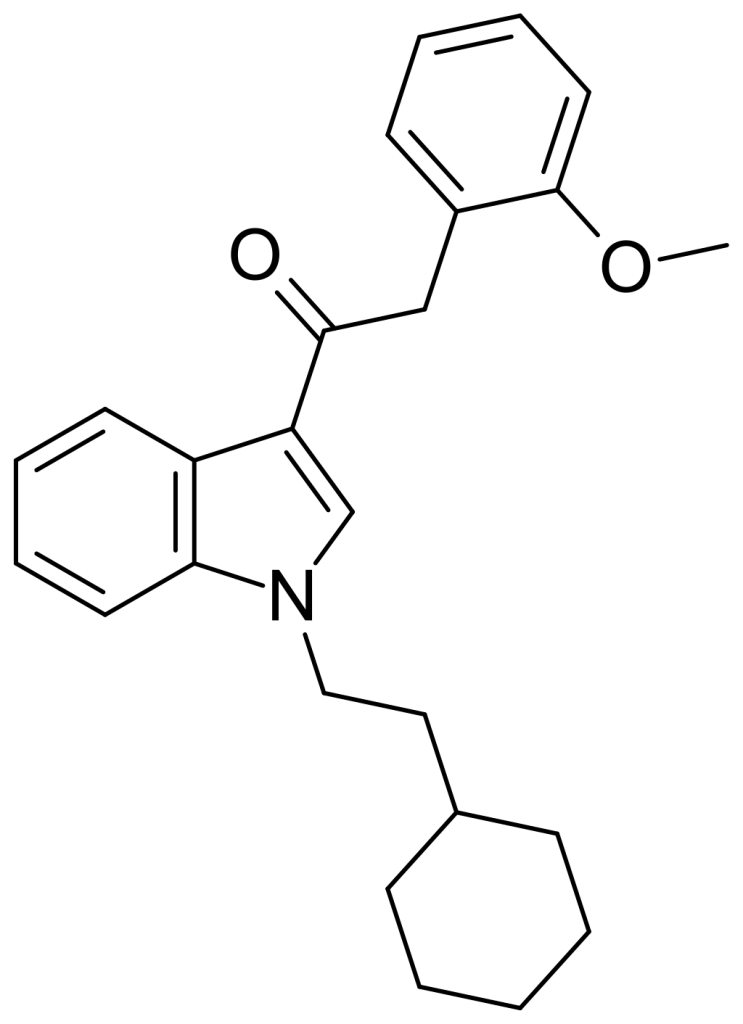
RCS-8, also known as 1-(2-cyclohexyl ethyl)-3-(2-methoxyphenylacetyl)indole and referred to as SR-18 and BTM-8, is a synthetic cannabinoid that has been identified as an ingredient in “herbal” synthetic cannabis mixtures. Structurally, it can be characterized as an analog of JWH-250, where the 1-pentyl group is replaced by 1-(2-cyclohexylethyl). As a result of this structural modification, RCS-8 is generally expected to have reduced potency compared to JWH-250, akin to the relationship between JWH-007 and its cyclohexylethyl analog.
While RCS-8 has not yet been reported in scientific or patent literature, anecdotal evidence suggests its recreational use in the United States. This prompted its specific inclusion in a proposed 2011 amendment to the Controlled Substances Act, which aimed to classify several synthetic drugs, including RCS-8, as Schedule I Controlled Substances. In the United States, all CB1 receptor agonists belonging to the 3-phenylacetylindole class, like RCS-8, are categorized as Schedule I Controlled Substances. This classification denotes their legal prohibition due to their potential for abuse and the absence of recognized medical applications.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1345970-42-4 [SciFinder] |
|---|---|
| ChemSpider | 24751863 |
| UNII | 11030I5W3R |
| CompTox Dashboard (EPA) | DTXSID30158821 |
| Chemical and physical data | |
| Formula | C25H29NO2 |
| Molar mass | 375.512 g·mol−1 |
FAQ
1. What is RCS-8?
RCS-8, also known as 1-(2-cyclohexyl ethyl)-3-(2-methoxyphenylacetyl)indole, is a synthetic cannabinoid used in “herbal” synthetic cannabis mixtures.
2. How does RCS-8 compare to other synthetic cannabinoids?
RCS-8 is structurally similar to JWH-250 but has a modification with a 1-(2-cyclohexyl ethyl) group instead of the 1-pentyl group, making it an analog of JWH-250. This structural change is expected to result in reduced potency compared to JWH-250.
3. What is its legal status in the United States?
In the United States, RCS-8 is classified as a Schedule I Controlled Substance. This categorization means it is prohibited due to its potential for abuse and the absence of recognized medical applications.
4. Has RCS-8 been studied scientifically or in patent literature?
While RCS-8 has not been reported in scientific or patent literature, there have been reports of its recreational use in the United States.
5. Why was RCS-8 listed explicitly in a proposed 2011 amendment to the Controlled Substances Act?
RCS-8 was included in a 2011 amendment to the Controlled Substances Act due to its reputed recreational use. The amendment aimed to classify several synthetic drugs, including RCS-8, as Schedule I Controlled Substances.
6. What are the potential effects and risks associated with RCS-8 use?
The specific effects and risks of RCS-8 use may vary, but as a synthetic cannabinoid, it may lead to altered perceptions, relaxation, and potential adverse reactions. The safety profile of RCS-8 is not well-established.
7. Is RCS-8 related to synthetic cannabis blends?
Yes, RCS-8 has been identified as an ingredient in “herbal” synthetic cannabis blends, making it part of the evolving landscape of synthetic cannabinoids.
8. Where can I find more information about RCS-8?
To gather more information about RCS-8, you can refer to credible scientific sources, drug regulation agencies, and educational websites. It’s crucial to rely on reliable sources when seeking information about substances like RCS-8.
References
- In 1994, an important milestone was reached when Huffman JW, Dai D, Martin BR, and Compton DR ventured into the world of “Cannabimimetic Indoles.” Their work encompassed the design, synthesis, and pharmacology of these indoles, shedding light on their potential applications and properties. This research was documented in Bioorganic & Medicinal Chemistry Letters, Volume 4, Issue 4, Pages 563–566. [DOI: 10.1016/S0960-894X(01)80155-4]
- The Synthetic Drug Control Act of 2011 stands as a significant legislative development in the United States. This act was enacted to address the growing concerns related to synthetic drugs, their composition, and the associated risks. It aimed to control and regulate various synthetic substances, particularly those with psychoactive effects.
- A crucial element in understanding drug regulation in the United States is 21 U.S.C. § 812. This federal statute outlines the schedules of controlled substances, categorizing them based on their recognized medical uses, potential for abuse, and overall legal status. This legal framework plays a pivotal role in the regulation and control of various substances within the country.