Beautiful Plants For Your Interior
Summary
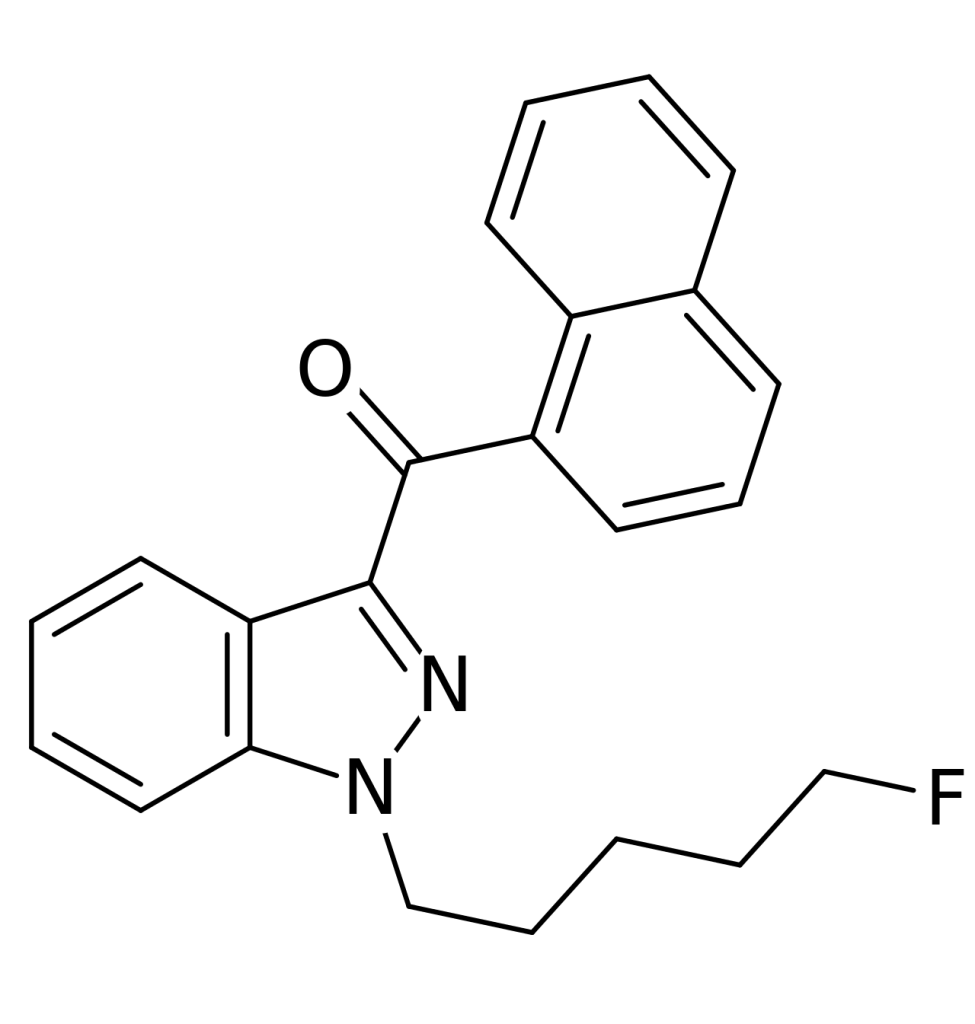
THJ-2201 is a synthetic cannabinoid based on the indazole structure, and it is believed to function as a potent agonist for the CB1 receptor. This compound has been available for purchase online as a designer drug. Structurally, THJ-2201 is akin to AM-2201, with the central indole ring substituted by an indazole ring.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 1801552-01-1 |
|---|---|
| PubChem CID | 91864533 |
| ChemSpider | 30646749 |
| UNII | D7P051IW0T |
| CompTox Dashboard (EPA) | DTXSID901010029 |
| Chemical and physical data | |
| Formula | C23H21FN2O |
| Molar mass | 360.432 g·mol−1 |
Pharmacology
THJ-2201 exhibits full agonism, with a remarkable binding affinity of 1.34nM at CB1 and 1.32nM at CB2 cannabinoid receptors.
Side effects
Concerningly, THJ-2201 has been associated with at least one instance of hospitalization and fatality resulting from its use.
Legal status
Due to the considerable risks associated with its recreational use, THJ-2201 is categorized as a Schedule I controlled substance in the United States, and it is designated as an Anlage II controlled drug in Germany.
FAQ
1. What is THJ-2201?
- THJ-2201 is a synthetic cannabinoid known for its activity as a full agonist at CB1 and CB2 cannabinoid receptors. It has been associated with recreational use.
2. What is the binding affinity of THJ-2201 to CB1 and CB2 receptors?
- THJ-2201 exhibits a binding affinity of 1.34nM at CB1 and 1.32nM at CB2 receptors.
3. Are there any known side effects of THJ-2201?
- Yes, THJ-2201 has been linked to adverse health effects, including at least one hospitalization and a reported fatality due to its use.
4. What is the legal status of THJ-2201 in the United States?
- In the United States, THJ-2201 is classified as a Schedule I controlled substance. This categorization reflects its potential for abuse and associated health risks.
5. How is THJ-2201 regulated in Germany?
- THJ-2201 is designated as an Anlage II controlled drug in Germany, indicating a level of legal control due to its potential risks and effects.
6. Is THJ-2201 safe for recreational use?
- THJ-2201 is associated with significant health risks and has been linked to hospitalization and death. It is not considered safe for recreational use.
7. Is THJ-2201 used for any medical purposes?
- THJ-2201 is not approved for medical use and is primarily known for its recreational use as a designer drug.
8. Where can I find more information about THJ-2201 and its effects?
- For detailed information about THJ-2201, its effects, legal status, and potential health risks, consult reputable sources such as government health agencies, medical professionals, and substance abuse organizations.
References
- In a January 2016 study authored by Diao X, Wohlfarth A, Pang S, Scheidweiler KB, and Huestis MA, titled “High-Resolution Mass Spectrometry for Characterizing the Metabolism of Synthetic Cannabinoid THJ-018 and Its 5-Fluoro Analog THJ-2201 after Incubation in Human Hepatocytes,” the metabolic processes of synthetic cannabinoids THJ-018 and THJ-2201 in human hepatocytes were investigated. The study, featured in Clinical Chemistry, Volume 62, Issue 1, spans pages 157 to 169 and can be accessed through doi:10.1373/clinchem.2015.243535. The PMID is 26430074.
- In September 2014, Shevyrin V, Melkozerov V, Nevero A, Eltsov O, Morzherin Y, and Shafran Y conducted research in the field of synthetic cannabinoids. Their work titled “3-Naphthoylindazoles and 2-naphthoylbenzoimidazoles as novel chemical groups of synthetic cannabinoids: chemical structure elucidation, analytical characteristics and identification of the first representatives in smoke mixtures” was published in Forensic Science International, Volume 242, pages 72 to 80. The study can be accessed via doi:10.1016/j.forsciint.2014.06.022, and the PMID is 25036783.
- In August 2014, Nahoko Uchiyama, Yoshihiko Shimokawa, Maiko Kawamura, Ruri Kikura-Hanajiri, and Takashi Hakamatsuka explored various designer drugs, including benzofuran derivative 2-(2-ethylaminopropyl)benzofuran (2-EAPB) and synthetic cannabinoids. Their findings were documented in “Chemical analysis of a benzofuran derivative, 2-(2-ethylaminopropyl)benzofuran (2-EAPB), eight synthetic cannabinoids, five cathinone derivatives, and five other designer drugs newly detected in illegal products,” published in Forensic Toxicology, Volume 32, Issue 2, spanning pages 266 to 281. The study can be accessed via doi:10.1007/s11419-014-0238-5, and S2CID is 11873421.
- In a 2016 study by Diao X, Scheidweiler KB, Wohlfarth A, Zhu M, Pang S, and Huestis MA, titled “Strategies to distinguish new synthetic cannabinoid FUBIMINA (BIM-2201) intake from its isomer THJ-2201: metabolism of FUBIMINA in human hepatocytes,” the metabolism of FUBIMINA and THJ-2201 in human hepatocytes was examined. The study, featured in Forensic Toxicology, Volume 34, Issue 2, covers pages 256 to 267. It can be accessed through doi:10.1007/s11419-016-0312-2. The study is also available in PMC with reference to PMC 4971051, and the PMID is 27547265.
- Information source: “THJ-2201”. Provided by Cayman Chemical. Date of retrieval: July 21, 2015.
- In a study published in July 2016 by Hess C, Schoeder CT, Pillaiyar T, Madea B, and Müller CE, titled “Pharmacological evaluation of synthetic cannabinoids identified as constituents of spice,” the pharmacological properties of synthetic cannabinoids present in spice products were evaluated. The study was published in Forensic Toxicology, Volume 34, Issue 2, spanning pages 329 to 343. It can be accessed via doi:10.1007/s11419-016-0320-2. The study is also available in PMC with reference to PMC 4929166, and the PMID is 27429655.
- In a July 2015 article authored by Trecki J, Gerona RR, and Schwartz MD, titled “Synthetic Cannabinoid-Related Illnesses and Deaths,” the relationship between synthetic cannabinoids and health issues was explored. The article was featured in The New England Journal of Medicine, Volume 373, Issue 2, on pages 103 to 107. It can be accessed via doi:10.1056/NEJMp1505328, and the PMID is 26154784.
- The “N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-(cyclohexylmethyl)-1H-indazole-3-carboxamide (AB-CHMINACA), N-(1-amino-3-methyl-1-oxobutan-2-yl)-1-pentyl-1H-indazole-3-carboxamide (AB-PINACA) and 1-(5-fluoropentyl)-1H-indazol-3-ylmethanone (THJ-2201): Background Information and Evaluation of ‘Three Factor Analysis’ (Factors 4, 5, and 6) for Temporary Scheduling” is a document published by the Drug and Chemical Evaluation Section of the Office of Diversion Control, Drug Enforcement Administration, in December 2014.
- The “Schedules of controlled substances: temporary placement of three synthetic cannabinoids into schedule I. Final order” was issued by the Drug Enforcement Administration, Department of Justice, in January 2015, and was published in the Federal Register, Volume 80, Issue 20, on pages 5042 to 5047. The PMID for this publication is 25730924.
- The legal status and classification of THJ-2201 in Germany are detailed in the “Gesetz über den Verkehr mit Betäubungsmitteln (Betäubungsmittelgesetz – BtMG) Anlage II (zu § 1 Abs. 1) (verkehrsfähige, aber nicht verschreibungsfähige Betäubungsmittel).” This information was retrieved on July 9, 2015.