Beautiful Plants For Your Interior
Summary
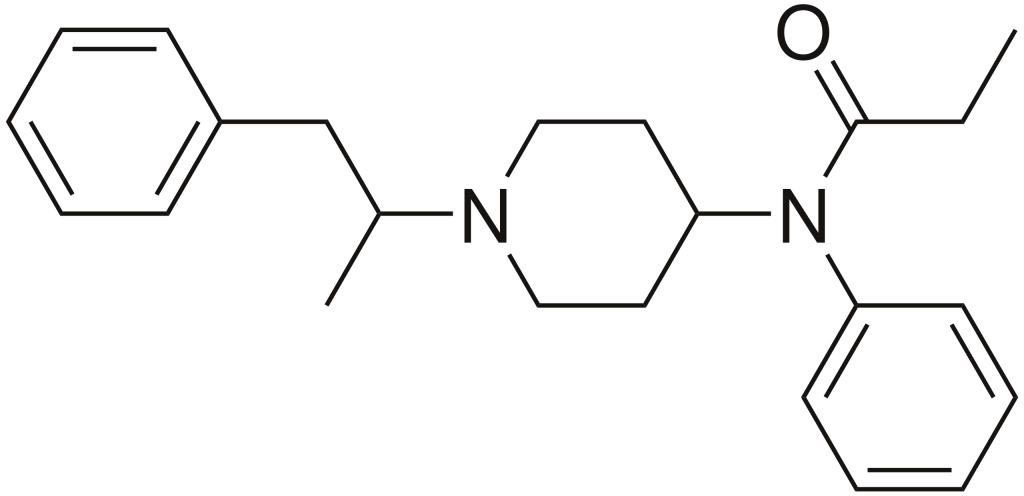
α-Methylfentanyl, also known as alpha-Methylfentanyl, is an opioid analgesic that belongs to the fentanyl analog family. This substance is occasionally marketed under the street name “China White.”
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 79704-88-4 |
|---|---|
| PubChem CID | 62281 |
| DrugBank | DB01557 |
| ChemSpider | 56081 |
| UNII | 8FU589W85C |
| CompTox Dashboard (EPA) | DTXSID60273955 |
| Chemical and physical data | |
| Formula | C23H30N2O |
| Molar mass | 350.506 g·mol−1 |
History
α-Methylfentanyl was initially synthesized by a team at Janssen Pharmaceutica during the 1960s. By 1976, it had started appearing as an additive mixed with heroin, and this combination was often referred to as “China White.” However, its notoriety grew in December 1979 when it was identified in the bodies of two drug overdose victims in Orange County, California. These individuals had exhibited symptoms of opiate overdose but tested negative for any known drugs of this class. Over the subsequent year, 13 more fatalities occurred, and α-Methylfentanyl was eventually determined to be the causative agent.
In a swift response to its emergence on the streets, α-Methylfentanyl was placed on the U.S. Schedule I list in September 1981, only two years after it was first encountered. Nonetheless, the development of various analogs of fentanyl continued to evolve rapidly. These analogs included para-fluoro fentanyl, α-methylacetylfentanyl, 3-methylfentanyl (a highly potent variant), and numerous others like β-hydroxyfentanyl, ohmefentanyl, β-hydroxythiofentanyl, and β-hydroxy-4-methylfentanyl. This proliferation of novel narcotic drugs prompted the implementation of the Federal Analog Act, which sought to regulate entire drug families based on their structural similarity rather than scheduling each new analog individually upon its appearance.
The chemical structure of fentanyl has served as a foundation in modern chemistry for the discovery and vocabulary of many new fentanyl analogs, commonly referred to as catalogs.
In 1991, a group of Russian chemistry students devised a simplified synthesis method that involved phosgene instead of phenethylamine. Consequently, the drug’s abuse became widespread, contributing to a significant portion of overdoses in the Moscow region. α-Methylfentanyl gained notoriety for its low safety profile, which eventually led to a decline in its production.
Effects
α-Methylfentanyl exhibits effects similar to those of fentanyl. While it is less potent by weight, this is attributed to reduced binding affinity with its target site. However, it possesses a longer duration of action because the α-methyl group interferes with the binding of metabolic enzymes that break down the drug. Remarkably, the discovery of the influence of the α-methyl group on fentanyl marked a significant moment when clandestine recreational drug research had a practical impact on scientific research.
Given that fentanyl itself is highly potent and notorious for causing fatal overdoses when misused, and it has a concise duration of action, often requiring users to administer doses hourly, α-Methylfentanyl might present several advantages over the parent compound as a recreational drug. Side effects of fentanyl analogs, including α-Methylfentanyl and 3-methylfentanyl, closely resemble those of fentanyl itself. These side effects encompass itching, nausea, and the potential for severe respiratory depression, particularly in cases of overdoses or when combined with other substances, such as benzodiazepines, which can be life-threatening.
Fentanyl analogs like α-Methylfentanyl and 3-methylfentanyl are frequently employed as additives, in small quantities, in conventional heroin stamps and packages. This practice enhances their potency and profitability when mixed with heroin, as it increases the retail price and the power per unit, thereby making them more lucrative in the illicit drug market.
FAQ
1. What is α-Methylfentanyl?
α-Methylfentanyl is an opioid analgesic that is an analog of fentanyl. It is sometimes associated with street names like “China White.”
2. What are the effects of α-Methylfentanyl?
α-Methylfentanyl produces results similar to fentanyl, including pain relief and euphoria. It is known for its longer duration of action compared to fentanyl.
3. How does α-Methylfentanyl work?
α-Methylfentanyl works by binding to opioid receptors in the brain, similar to other opioids. It interferes with binding to metabolic enzymes, which contributes to its longer duration of action.
4. Is α-Methylfentanyl more potent than fentanyl?
No, α-Methylfentanyl is generally less potent by weight compared to fentanyl due to reduced binding affinity. However, its longer duration of action can make it attractive to specific users.
5. What are the side effects of α-Methylfentanyl?
Side effects are similar to those of fentanyl and may include itching, nausea, and respiratory depression. Overdoses or improper drug combinations, such as with benzodiazepines, can lead to life-threatening respiratory depression.
6. Why is α-Methylfentanyl used as an additive in heroin?
α-Methylfentanyl and similar analogs are often used as “cut” or additives in small amounts in heroin products. This practice increases the potency and profitability of the heroin, which benefits dealers by raising the retail price and force per unit.
7. Is α-Methylfentanyl safe to use?
No, like other potent opioids, α-Methylfentanyl carries significant risks, significantly when misused. It can lead to severe health complications, addiction, and overdose. It is not safe for recreational use.
8. Is α-Methylfentanyl legal?
In many countries, including the United States, α-Methylfentanyl is classified as a controlled substance and is illegal to manufacture, distribute, or possess without proper authorization.
9. What is the history of α-Methylfentanyl?
α-Methylfentanyl was first discovered in the 1960s by a team at Janssen Pharmaceutica. It gained notoriety when it began appearing mixed with heroin as an additive in the 1970s.
10. Can α-Methylfentanyl be used for medical purposes?
No, α-Methylfentanyl has no recognized medical use and is not approved for medical treatment. It is considered a dangerous and illicit substance.
Please note that α-Methylfentanyl is a potent and dangerous drug with a high risk of addiction and overdose. This FAQ is provided for informational purposes, and the use of such substances is strongly discouraged.
References
- US Patent 3164600: This patent document is a crucial reference for understanding the early history and development of certain substances, potentially shedding light on their origin and intended use.
- Kram TC, Cooper DA, Allen AC (1981): This source provides insights “Behind the identification of China White,” which may offer details about the emergence of this specific drug mixture.
- Gillespie TJ, Gandolfi AJ, Davis TP, Morano RA (1982): This publication focuses on the “Identification and quantification of alpha-methylfentanyl in post mortem specimens,” providing essential information about the drug’s presence in post-mortem cases.
- Henderson GL (1988): In this source, “Designer Drugs: Past History and Future Prospects” are discussed. This is significant in understanding the historical context and potential future trends in designer drugs.
- CHAZAN, GUY (May 27, 1993): This news article, titled “The trade in illegal narcotics looks set to mushroom,” offers a perspective on the illegal narcotics trade, which may indirectly relate to α-Methylfentanyl and similar substances.
- Ohmefentanyl: While not directly related to α-Methylfentanyl, information about Ohmefentanyl can provide a broader understanding of fentanyl analogs and their variations.