Beautiful Plants For Your Interior
Summary
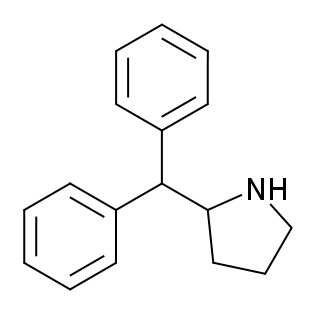
2-Diphenylmethylpyrrolidine, commonly known as Desoxy-D2PM or 2-benzhydrylpyrrolidine, is a potent psychoactive stimulant. It is structurally related to diphenylprolinol (D2PM) and shares similarities with desoxypipradrol (2-DPMP), both of which function as inhibitors of norepinephrine and dopamine reuptake (NDRIs). Desoxy-D2PM is often marketed as a designer drug and has been involved in the production of legal highs. It has been sold under various names, including A3A New Generation, A3A Methano, and Green Powder. It has been associated with adverse effects such as hallucinations, aggressive behaviour, pupil dilation, rapid heartbeat, and elevated blood pressure.
Studies suggest that Desoxy-D2PM can produce psychotropic effects similar to other stimulants but with a more prolonged duration of action.
Desoxy-D2PM has two enantiomers, and they are employed in their pure form in industrial settings as chiral derivatizing agents during chemical synthesis.
On November 4, 2010, the UK Home Office implemented an importation ban on 2-diphenylmethylpyrrolidine based on a recommendation from the Advisory Council on the Misuse of Drugs (ACMD). Initially set to become a Class B drug on March 28, 2012, this classification was postponed due to concerns over the inclusion of two steroids in the bill that were subsequently recommended to remain uncontrolled. Eventually, on June 13, 2012, Desoxy-D2PM was classified as a Class B drug and placed in Schedule I.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 119237-64-8 |
|---|---|
| PubChem CID | 1295 |
| ChemSpider | 1256 |
| UNII | UH7U0MAY6C |
| CompTox Dashboard (EPA) | DTXSID80922897 |
| ECHA InfoCard | 100.162.251 |
| Chemical and physical data | |
| Formula | C17H19N |
| Molar mass | 237.339 g·mol−1 |
Legal status
| Legal status | |
|---|---|
| Legal status | DE: NpSG (Industrial and scientific use only) UK: Class B |
FAQ
1. What is 2-Diphenylmethylpyrrolidine (Desoxy-D2PM)?
- Desoxy-D2PM, also known as 2-benzhydryl pyrrolidine, is a powerful psychoactive stimulant drug. It shares structural similarities with other stimulants like diphenylprolinol (D2PM) and desoxypipradrol (2-DPMP).
2. How does Desoxy-D2PM work?
- Desoxy-D2PM acts as a norepinephrine-dopamine reuptake inhibitor (NDRI). This means it affects the reabsorption of neurotransmitters, leading to increased levels of norepinephrine and dopamine in the brain.
3. What are the common street names for Desoxy-D2PM?
- Desoxy-D2PM has been sold under various names, including A3A New Generation, A3A Methano, and Green Powder.
4. What are the effects of Desoxy-D2PM?
- Desoxy-D2PM can produce stimulant effects, such as increased energy, alertness, and sometimes hallucinations. However, it is also associated with adverse effects like aggressive behaviour, dilated pupils, tachycardia (rapid heartbeat), and high blood pressure.
5. Is Desoxy-D2PM legal?
- The legal status of Desoxy-D2PM varies by country. In the UK, it was classified as a Class B drug and placed in Schedule I on June 13, 2012. It is considered illegal in many other jurisdictions as well.
6. Can Desoxy-D2PM be used for legitimate purposes?
- Desoxy-D2PM does not have approved medical uses and is primarily marketed and used as a designer drug.
7. Are there different forms or enantiomers of Desoxy-D2PM?
- Yes, Desoxy-D2PM has two enantiomers. In some industrial applications, these enantiomers are used in their purified form as chiral derivatizing agents during chemical synthesis.
8. Are there any safety concerns associated with Desoxy-D2PM use?
- Yes, using Desoxy-D2PM can be dangerous and is associated with a range of health risks, including the potential for addiction and adverse physiological and psychological effects.
9. Can Desoxy-D2PM be detected in drug tests?
- Desoxy-D2PM may not be included in standard drug tests, but specialized tests can detect its presence.
10. Where can I find more information about Desoxy-D2PM?
- Given the legal and health risks associated with Desoxy-D2PM, it is advisable to seek information from reputable sources, such as government health agencies or addiction support organizations. Additionally, consulting with healthcare professionals or substance abuse specialists is recommended if you have concerns about Desoxy-D2PM or similar substances.
References
- De Paoli G, Brandt SD, and Pounder DJ conducted an analytical study in December 2011, focusing on the characterization and rapid determination of 2-(diphenylmethyl)pyrrolidine in blood. Their research, published in the Journal of Chromatography. B, was aimed at providing insights into this compound and its presence in internet products (doi: 10.1016/j.jchromb.2011.10.014; PMID 22055832).
- For information in Italian, the Dipartimento Politiche Antidroga in Rome archived data related to “2-(Diphenylmethyl)pyrrolidine (desoxy-D2PM)” in a PDF format. This document was accessible until February 27, 2013, and retrieved on February 10, 2012.
- DC Shanie Nayar provided information about “Green Powder called A3A Methano” in a PDF format, with the document being archived until February 27, 2013. This resource was retrieved on February 10, 2012.
- In July 2012, Coppola M and Mondola R discussed research chemicals marketed as legal highs, specifically focusing on pipradrol derivatives, in Toxicology Letters (doi: 10.1016/j.toxlet.2012.04.019; PMID 22564760).
- Bertelsen S, Halland N, Bachmann S, Marigo M, Braunton A, and Jørgensen KA conducted a study in October 2005, exploring organocatalytic asymmetric alpha-bromination of aldehydes and ketones. This research was published in Chemical Communications (doi: 10.1039/b509366j; PMID 16193126).
- The UK Home Office implemented an import ban on the psychoactive drug and archived information regarding it. This information can be accessed through the provided link (Archived 29 August 2012 at the Wayback Machine).
- On January 27, 2012, the UK Home Office issued “The Misuse of Drugs Act 1971 (Amendment) Order 2012” in a PDF format, which can be referred to for legislative details.
- The UK Home Office accepted advice from the Advisory Council on the Misuse of Drugs (ACMD) to schedule D2PM, 2-DPMP, and phenzepam. This decision was communicated in a document available in PDF format (Archived from the original on 6 April 2012).
- The ACMD provided further advice on the classification of two steroidal substances in a letter dated February 14, 2012. This letter is accessible in PDF format (Archived from the original on 5 April 2012).
- The UK Home Office announced a change to the Misuse of Drugs Act 1971 on June 7, 2012, regarding the control of pipradrol-related compounds and phenazepam. Additional details can be found in the provided link (Archived from the original on 30 October 2012).