Beautiful Plants For Your Interior
Summary
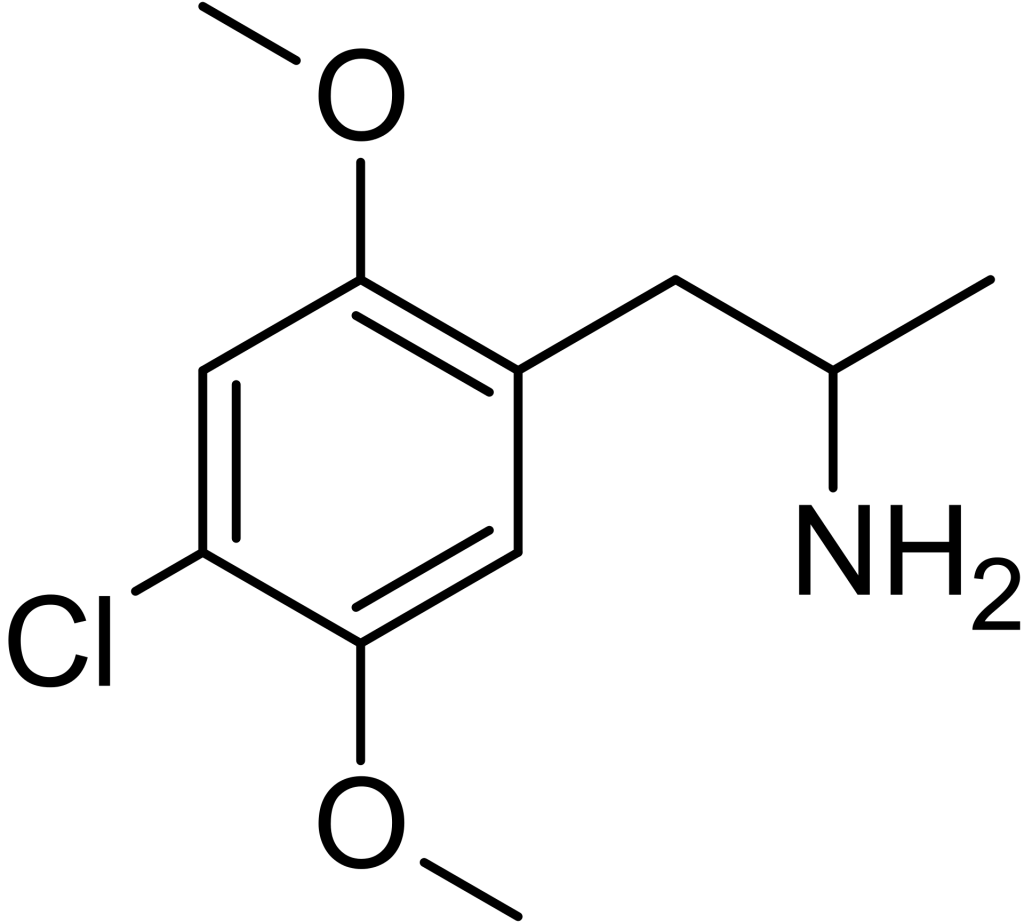
2,5-Dimethoxy-4-chloroamphetamine (DOC) belongs to the phenethylamine and amphetamine chemical groups and is recognized as a psychedelic substance. Its initial synthesis is attributed to Alexander Shulgin and is documented in detail within his seminal work “PiHKAL” (Phenethylamines I Have Known And Loved).
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 123431-31-2 |
|---|---|
| PubChem CID | 542036 |
| ChemSpider | 472008 |
| UNII | 8Y1C6K0PCA |
| ChEMBL | ChEMBL8100 |
| CompTox Dashboard (EPA) | DTXSID60894189 |
| ECHA InfoCard | 100.215.939 |
| Chemical and physical data | |
| Formula | C11H16ClNO2 |
| Molar mass | 229.70 g·mol−1 |
Chemistry
DOC falls within the category of substituted alpha-methylated phenethylamines, a well-known subgroup of compounds commonly referred to as amphetamines. The phenethylamine analog without the alpha-methyl group is recognized as 2C-C. Notably, DOC possesses a stereocenter, with (R)-(−)-DOC identified as the more potent stereoisomer.
Pharmacology
DOC functions as a discerning partial agonist at the 5-HT2A and 5-HT2C receptors. Its psychedelic impacts primarily manifest through its interactions with the 5-HT2A receptor.

Dosage
The standard dosage for DOC typically spans from 0.5 to 7.0 mg, with the lower range triggering threshold effects and the higher content inducing notably intense effects. The drug generally takes effect within 1 to 3 hours, reaching its peak and plateau phase at 4 to 8 hours, followed by a gradual descent with lingering stimulation lasting between 9 to 20 hours. Post-effects may extend well into the following day.
Effects
In contrast to basic amphetamines, DOC is recognized as a substance that notably impacts the cognitive and perceptual functions of the brain. Its most potent reported effects are enhanced visual and auditory perceptions, heightened sensitivity to sound and motion, and a sense of euphoria. In his autobiography “PiHKAL,” Alexander Shulgin characterized DOC as an “archetypal psychedelic” (#64), attributing to it a comprehensive spectrum of vivid visual, auditory, physical, and mental effects characterized by a profound clarity as well as overwhelming, humbling, and intertwining experiences.
Dangers
Information regarding the toxicity of DOC remains scarce. However, in April 2013, a reported case linked a fatality to the use of DOC, although the source did not clearly define the specific role of the drug in the death. Additionally, in 2014, an end was attributed directly to DOC as the solitary causative factor, with the autopsy revealing pulmonary edema and a subgaleal hemorrhage.
Detection in biological specimens
To confirm cases of poisoning in hospitalized individuals or provide evidence in medicolegal investigations, DOC can be measured in blood, plasma, or urine using gas chromatography-mass spectrometry or liquid chromatography-mass spectrometry. Individuals engaging in recreational use may exhibit blood or plasma DOC concentrations ranging from 1 to 10 μg/L. For intoxicated patients, concentrations may exceed 20 μg/L, while victims of acute overdosage may show concentrations surpassing 100 μg/L.
Popularity
Although a rarity in the black market, the global trade of DOC has been facilitated by exclusive “Grey Market” Research Chemical suppliers, allowing for widespread distribution in bulk over several years. Incidences of DOC sold on blotting paper and in capsules were recorded in the later months of 2005 and 2007. Notably, a DEA Microgram report from December 2007 detailed a seizure by the Concord Police Department in Contra Costa County, California, revealing that what was initially suspected to be LSD “blotter acid” turned out to be DOC upon thorough analysis. Unscrupulous dealers have been known to misrepresent DOC as LSD, posing significant risks, as DOC lacks the established safety profile of LSD. This misrepresentation is particularly hazardous, especially for individuals with hypertension, as amphetamine compounds can lead to sudden spikes in systolic blood pressure, potentially exacerbating pre-existing health concerns.
Drug prohibition laws
Canada
DOC is classified as a Schedule 1 substance[9] due to its categorization as an amphetamine analog. The Safe Streets Act led to amendments in the CDSA, resulting in the reclassification of amphetamines from Schedule 3 to Schedule 1.
Australia
It is not formally scheduled, but it can potentially be regulated as a Schedule II substance as an analog of DOB.
China
As of October 2015, DOC is designated a controlled substance in China.
New Zealand
Scheduled accordingly.
Denmark
DOC was included in the Schedule I controlled substances list in Denmark effective from 8.4.2007.
Germany
Listed as a controlled substance in Anlage I since 22.1.2010.[12]
Sweden
Sveriges Riksdag incorporated DOC into Schedule I (“substances, plant materials, and fungi typically without medical use”) as narcotics in Sweden as of Aug 30, 2007. The Medical Products Agency published this in their regulation LVFS 2007:10, specified as DOC, 4-klor-2,5-dimetoxi-amfetamin.[14] Initially classified as a “health hazard” by the health ministry Statens folkhälsoinstitut under the Act on the Prohibition of Certain Goods Dangerous to Health as of Jul 1, 2004, in their regulation SFS 2004:486, identified as 4-klor-2,5-dimetoxiamfetamin (DOC).
United Kingdom
Categorized as Class A.
United States
DOC is not classified or controlled at the federal level in the United States [16], but the Department of Justice views it as an analog of DOB[17]. Therefore, its possession or sale could be prosecuted under the Federal Analogue Act. The analogs DMA, DOB, and DOM are designated as Schedule I controlled substances in the United States.
US State of Florida
DOC is a Schedule I controlled substance in Florida, rendering it illegal to purchase, sell, or possess.
United Nations
In December 2019, the UNODC proposed scheduling recommendations that would place DOC into Schedule I alongside several other research chemicals.
FAQ
- What is 2,5-Dimethoxy-4-Chloroamphetamine (DOC)?
- DOC, short for 2,5-Dimethoxy-4-Chloroamphetamine, is a psychedelic substance belonging to the amphetamine and phenethylamine chemical classes. It is known for its hallucinogenic properties.
- How does DOC affect the body and mind?
- DOC primarily acts as a selective partial agonist at the 5-HT2A and 5-HT2C serotonin receptors, contributing to its psychedelic effects. Users commonly experience heightened sensory perception, open and closed-eye visuals, and increased awareness of sound and movement. It is often described as an “archetypal psychedelic.”
- What is the typical dosage for DOC?
- The average dose of DOC ranges from 0.5 to 7.0 mg. Lower doses may produce threshold effects, while higher doses can lead to intensely psychedelic solid experiences. The onset of the drug is generally within 1 to 3 hours, with peak effects occurring at 4 to 8 hours. The experience gradually tapers down, with residual stimulation lasting up to 20 hours.
- Is DOC related to other substances?
- DOC belongs to the substituted alpha-methylated phenethylamine class. It is chemically similar to other psychedelic compounds but possesses its unique effects.
- What are the risks and side effects of using DOC?
- The toxicity of DOC is not well-documented, and there have been reports of fatalities associated with its use. High doses may lead to issues like arterial spasms. Additionally, misrepresenting DOC as LSD by unscrupulous dealers can be particularly dangerous, especially for individuals with hypertension.
- Is DOC legal in my country?
- The legal status of DOC varies by country. It may be classified as a controlled substance, an analog of other substances, or entirely unscheduled. It’s essential to research and understand the specific regulations in your area.
- How can DOC be detected in the body?
- DOC can be quantitated in blood, plasma, or urine using gas chromatography-mass or liquid chromatography-mass spectrometry. Concentrations can vary depending on the individual’s use, with recreational users typically showing lower levels than intoxicated or overdose cases.
- Do international drug control agencies regulate DOC?
- DOC is subject to control under international drug control treaties. The UNODC announced scheduling recommendations in December 2019 to place DOC into Schedule I, along with other research chemicals.
References
- Anvisa (2023-07-24): “RDC Nº 804 – Catalog of Controlled Substances” [Collegiate Board Resolution No. 804 – Lists of Controlled Substances] (in Brazilian Portuguese). Published in Diário Oficial da União (released on 2023-07-25). This source is archived and can be accessed until 2023-08-27. The retrieval date is also 2023-08-27.
- Shulgin, A. & Shulgin, A. (September 1991). “PiHKAL: A Chemical Love Story.” Published in the United States by Transform Press, page 978. The ISBN is 0-9630096-0-5. The source is archived and can be accessed until 3 January 2010. The retrieval date is 30 November 2009.
- “Erowid DOC Vault: Dosage.” This source is archived and can be accessed until 2 May 2008. The retrieval date is 22 April 2008.
- “Erowid Online Books: “PiHKAL” – #64 DOC.” This source is archived and can be accessed until 3 August 2018. The retrieval date is 17 November 2005.
- Bucks, J. (25 April 2013). “Moment of madness: rare drug implicated in student death.” Published in The Saint. The source is archived and can be accessed until 28 April 2013. The retrieval date is 25 April 2013.
- Barnett, R.Y., Baker, D.D., Kelly, N.E., McGuire, C.E., Fassette, T.C., & Gorniak, J.M. (October 2014). “A fatal intoxication of 2,5-dimethoxy-4-chloroamphetamine: a case report.” Published in the Journal of Analytical Toxicology, volume 38, issue 8, pages 589–91. doi:10.1093/jat/bku087. PMID 25217551.
- Baselt, R.C. (2014). “Disposition of toxic drugs and chemicals in man.” Published in Seal Beach, California by Biomedical Publications, page 2173. The ISBN is 978-0-9626523-9-4.
- “mg1207” (PDF). This source is archived and can be accessed until 2012-10-17. The retrieval date is 2013-08-13.
- “Schedule I.” Controlled Drugs and Substances Act. Canadian Legal Information Institute. The information is up to date as of 2022-03-31. The source is archived and can be accessed until 2022-03-31. The retrieval date is also 2022-03-31.
- “Controlled Drugs and Substances Act: Definitions and interpretations.” Canadian Legal Information Institute.
- “Backgrounder: The Safe Streets and Communities Act Four Components Coming Into Force.” Canadian Department of Justice. This source is archived and can be accessed until 2012-10-18.
- “Erowid DOC Vault: Legal Status.” This source is archived and can be accessed until 1 May 2008. The retrieval date is 22 April 2008.
- “关于印发《非药用类麻醉药品和精神药品列管办法》的通知” (in Chinese). Published by the China Food and Drug Administration on 27 September 2015. The source is archived and can be accessed until 1 October 2015. The retrieval date is also 1 October 2015.
- “Läkemedelsverkets föreskrifter – LVFS och HSLF-FS | Läkemedelsverket” (PDF).
- “Förordning om ändring i förordningen (1999:58) om förbud mot vissa hälsofarliga varor” (PDF) (in Swedish). Published on 27 May 2004. The source is archived and can be accessed until 15 September 2018.
- “PART 1308 – Section 1308.11 Schedule I.” This information is available at www.deadiversion.usdoj.gov and is archived until 27 August 2009. The retrieval date is 31 March 2018.
- “DEA Resources, Microgram, October 2007” (PDF). This source is archived and can be accessed until 2012-10-17. The retrieval date is 2012-10-03.
- “The 2017 Florida Statutes – Title XLVI – CRIMES – Chapter 893 – DRUG ABUSE PREVENTION AND CONTROL.” This information can be found at leg.state.fl.us. The retrieval date is 31 March 2018.
- “December 2019 – WHO: World Health Organization recommends 12 NPS for scheduling.”