Beautiful Plants For Your Interior
Summary
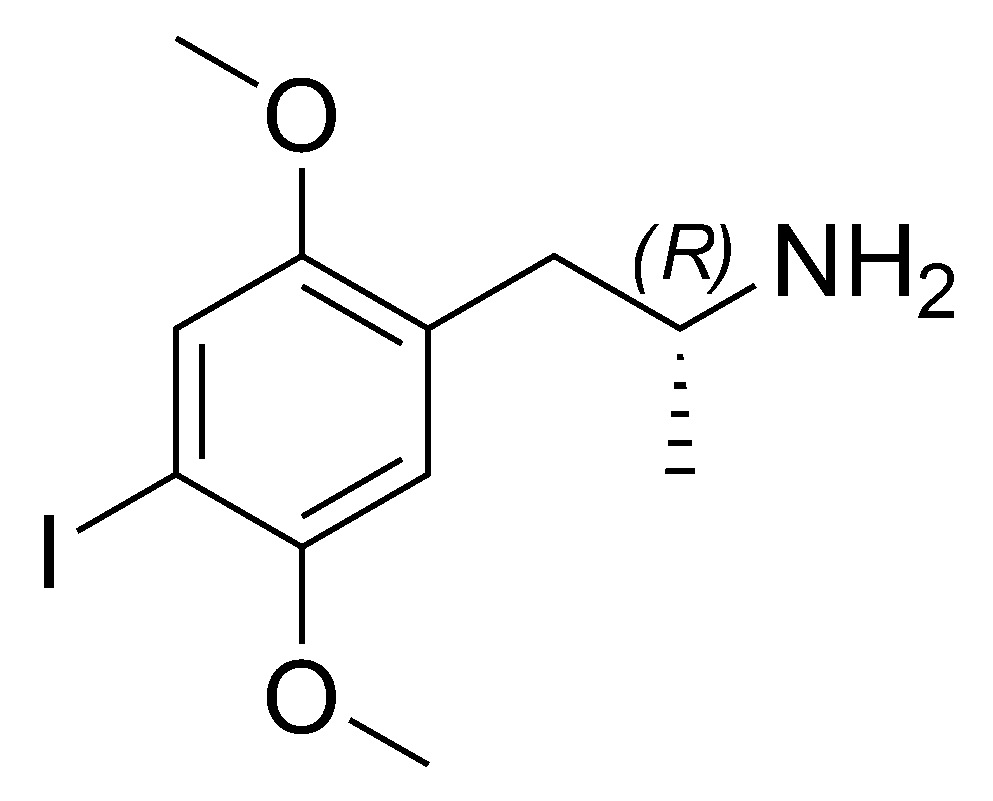
2,5-Dimethoxy-4-iodoamphetamine (DOI) is categorized as a psychedelic substance and a form of substituted amphetamine. Unlike other substituted amphetamines, DOI’s primary effects do not primarily manifest as stimulants. The R-(−)-DOI stereoisomer demonstrates more excellent activity with a stereocenter. In neuroscience research, [125I]-R-(−)-DOI is employed as a radioligand, indicating the presence of 5-HT2A serotonin receptors. Comparisons between DOI’s effects and LSD have been made, although seasoned users can discern distinct differences. Aside from its prolonged duration, the DOI trip is often noted for its heightened energy compared to an LSD trip, alongside a more pronounced body load and a distinct subjective visual encounter. Subsequent effects encompass residual stimulation and challenges with sleep, which, depending on the dosage, might endure for several days. While not prevalent, DOI is occasionally vented as a substitute for LSD or even deceptively marketed as LSD. This action can be challenging given that DOI does not possess the same well-established safety profile as LSD.
Research
According to recent findings, the introduction of (R)-DOI is observed to impede pulmonary inflammation, excessive mucus production, and heightened airway sensitivity while also deactivating crucial genes within the lung’s immune response. These effects contribute to preventing allergic asthma development in a mouse model.
In a surprising discovery, several hallucinogens functioning as 5-HT2A agonists, such as (R)-2,5-dimethoxy-4-iodoamphetamine DOI, TCB-2, LSD, and LA-SS-Az, have exhibited potent inhibition of TNF (tumor necrosis factor). Notably, DOI emerges as the most powerful, displaying TNF inhibition in the picomolar range, surpassing its hallucinogenic effects by an order of magnitude.
Pharmacology
| Receptor | Ki (racemic DOI)[9][failed verification] | Ki (R-DOI)[9][failed verification] | Ki (S-DOI)[9][failed verification] | Intrinsic activity[10] |
|---|---|---|---|---|
| 5-HT1A | 2355 nM | 3843 nM | ND | ND |
| 5-HT1B | 1261 nM | ND | ND | ND |
| 5-HT1D | 1241.3 nM | ND | ND | ND |
| 5-HT1E | 2970 nM | ND | ND | ND |
| 5-HT1F | 2125.44 nM | ND | ND | ND |
| 5-HT2A | 0.68 nM | 0.65 nM | 0.65 nM | Partial agonist. |
| 5-HT2B | 20.03 nM | 53.70318 nM | 28.183829 nM | Partial agonist/full agonist |
| 5-HT2C | 2.38 nM | 5.370318 nM | 8.317638 nM | Full agonist when coupled to phospholipase A. Partial agonist (intrinsic efficacy = 53%), when coupled to phospholipase C. |
| 5-HT5A | 1000 nM | ND | ND | ND |
| 5-HT6 | >10000 nM | ND | ND | ND |
In cellular investigations, DOI has demonstrated a remarkably potent capability to inhibit tumor necrosis factor-alpha (TNF-alpha) inflammation at picomolar concentrations. TNF-alpha serves as a critical focus for research concerning degenerative ailments like rheumatoid arthritis and Alzheimer’s disease, where the disease progression involves tissue damage resulting from persistent inflammation. This notable characteristic could establish DOI and other 5-HT2A agonists as promising avenues for developing novel treatments for these conditions.
Moreover, DOI has exhibited the ability to induce rapid growth and reorganization of dendritic spines and synaptic connections with other neurons, processes recognized to underlie neuroplasticity.
History
Alexander Shulgin is credited with the initial synthesis of DOI. The radioactive iodine-125 variant of DOI intended for PET imaging was initially formulated within the research facilities of David E. Nichols.
In January 2007, British law enforcement officials disclosed that three young men had become unwell, allegedly due to the ingestion of DOI at a rave in Biggleswade, near Milton Keynes. They cautioned other individuals who had consumed the substance to seek medical aid promptly. This incident indicates that DOI has begun to be more commonly utilized as a recreational drug in the United Kingdom.
Legal status
Australia:
DOI does not appear on the list of prohibited substances according to the Standard for the Uniform Scheduling of Medicines and Poisons (SUSMP) in Australia.
Canada:
It is classified as a Schedule 1 substance in Canada because it is considered an analog of amphetamine. The Canadian Controlled Drugs and Substances Act (CDSA) was amended to implement the Safe Streets and Communities Act, moving amphetamines from Schedule 3 to Schedule 1.
Denmark:
DOI has been illegal in Denmark since 8 April 2007.
Sweden:
The Swedish Parliament, Sveriges Riksdag, included DOI in schedule I, categorized as “substances, plant materials, and fungi which normally do not have medical use,” as narcotics in Sweden from August 30, 2007. The Medical Products Agency published this information in their regulation LVFS 2007:10, listed as DOI, 4-jod-2,5-dimetoxi-amfetamin.
United States:
DOI is not officially scheduled in the United States. However, DOI would probably be deemed an analog of DOB, potentially subjecting sales or possession to prosecution under the Federal Analogue Act. DOI is commonly utilized in animal and in vitro research. Scheduling DOI could present challenges for medical researchers.
US State of Florida:
DOI is classified as a Schedule I controlled substance in Florida.
FAQ
- What is 2,5-Dimethoxy-4-iodoamphetamine (DOI)? 2,5-Dimethoxy-4-iodoamphetamine (DOI) is a psychedelic compound that falls within the amphetamine class, known for its hallucinogenic effects and distinct chemical structure.
- Who discovered DOI? DOI was first synthesized and identified by renowned chemist Alexander Shulgin, known for his extensive research in the field of psychoactive substances.
- What are the effects of DOI? DOI is known to produce profound psychedelic effects, often characterized by altered perceptions, sensory distortions, and changes in thought patterns.
- Is DOI used for any medical purposes? DOI is primarily used in scientific research and is not approved for any medical applications. Studies have focused on its potential effects on specific receptors and its impact on certain biological processes.
- How is DOI regulated internationally? Regulations for DOI vary across different countries. While some nations have classified it as a controlled substance, others have not placed specific restrictions on its use.
- What are the potential risks associated with DOI use? The use of DOI, like other psychedelics, can pose various risks, including psychological distress, altered perception of reality, and potential long-term effects on mental health.
- Can DOI be used recreationally? DOI has been known to be used recreationally for its hallucinogenic properties. However, its use outside of controlled research settings can be hazardous and may lead to unpredictable and adverse effects.
- Is DOI legal in any country? DOI’s legal status varies by country. While some nations have strict regulations on its use, others may not have specific laws that pertain to DOI specifically.
References
- “D101 DOI hydrochloride, solid” – Information retrieved on April 13, 2008.
- Anvisa (2023-07-24). “RDC Nº 804 – Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial” [Collegiate Board Resolution No. 804 – Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Published in the Diário Oficial da União on July 25, 2023. The original document is archived until August 27, 2023, and the retrieval was conducted on August 27, 2023.
- Shulgin, A; Shulgin, A (1990). “#67 DOI”. PiHKAL: A Chemical Love Story. Published by Transform Press. The ISBN is 978-0963009609. The information was retrieved from the original source on October 27, 2014.
- “LSD Blotter Acid Mimics (Actually containing 4-IODO-2,5-DIMETHOXYAMPHETAMINE (DOI) and 4-CHLORO-2,5-DIMETHOXYAMPHETAMINE (DOC)) in Lantana, Florida.” DEA Microgram Bulletin. Published by the Office of Forensic Sciences, Drug Enforcement Administration, Washington, DC, in June 2008. The original document is archived until February 4, 2009, and the retrieval was conducted on February 12, 2009.
- “LSU Health New Orleans research finds psychedelic drug prevents asthma development in mice.” Source: EurekAlert!
- Miller KJ, Gonzalez HA (December 1998). “Serotonin 5-HT2A receptor activation inhibits cytokine-stimulated inducible nitric oxide synthase in C6 glioma cells.” Published in Annals of the New York Academy of Sciences, volume 861, issue 1, pages 169–73. doi:10.1111/j.1749-6632.1998.tb10188.x. PMID: 9928254. S2CID 23264746.
- Yu B, Becnel J, Zerfaoui M, Rohatgi R, Boulares AH, Nichols CD (November 2008). “Serotonin 5-hydroxytryptamine(2A) receptor activation suppresses tumor necrosis factor-alpha-induced inflammation with extraordinary potency.” Published in the Journal of Pharmacology and Experimental Therapeutics, volume 327, issue 2, pages 316–23. doi:10.1124/jpet.108.143461. PMID: 18708586. S2CID 25374241.
- Pelletier M, Siegel RM (December 2009). “Wishing away inflammation? New links between serotonin and TNF signaling.” Published in Molecular Interventions, volume 9, issue 6, pages 299–301. doi:10.1124/mi.9.6.5. PMC 2861806. PMID: 20048135.
- Roth, BL; Driscol, J (January 12, 2011). “PDSP Ki Database.” Source: Psychoactive Drug Screening Program (PDSP) by the University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved from the original source on November 8, 2013.
- Canal, CE; Morgan, D (July 2012). “Head-twitch response in rodents induced by the hallucinogen 2,5-dimethoxy-4-iodoamphetamine: a comprehensive history, a re-evaluation of mechanisms, and its utility as a model.” Published in Drug Testing and Analysis, volume 4, issues 7–8, pages 556–576. doi:10.1002/dta.1333. PMC 3722587. PMID: 22517680.
- Jones, KA; Srivastava, DP; Allen, JA; Strachan, RT; Roth, BL; Penzes, P (November 17, 2009). “Rapid modulation of spine morphology by the 5-HT2A serotonin receptor through kalirin-7 signaling.” Published in the Proceedings of the National Academy of Sciences, volume 106, issue 46, pages 19575–19580. doi:10.1073/pnas.0905884106. PMC 2780750. PMID: 19889983.
- “New drug alert as three taken ill.” Source: BBC News. Published on January 29, 2007.
- Gill, A (July 22, 2013). “POISONS STANDARD 2013” (PDF). Therapeutic Goods Administration. Australian Government Department of Health and Ageing. Retrieved on March 4, 2014.
- “Controlled Drugs and Substances Act: Legislative history · Schedule I · Section 19: Tramadol [Proposed]; Amphetamines.” Source: isomerdesign.com. Archived from the original, retrieved on March 31, 2022. Retrieved on November 27, 2012.
- “Controlled Drugs and Substances Act: Definitions and Interpretations.” Source: isomerdesign.com.
- Erowid DOC Vault: Legal Status. Retrieved from www.erowid.org.
- “Läkemedelsverkets föreskrifter – LVFS och HSLF-FS | Läkemedelsverket” (PDF).
- “PART 1308 – Section 1308.11 Schedule I.” Source: www.deadiversion.usdoj.gov. Archived from the original until August 27, 2009. Retrieved on December 17, 2014.
- Mario de la Fuente Revenga; Bohan Zhu; Christopher A. Guevara; George W. Huntley; Chang Lu; Javier González-Maeso (2021). “Prolonged epigenomic and synaptic plasticity alterations following single exposure to a psychedelic in mice.” Published in Cell Reports, volume 37, issue 3, article 109836. doi:10.1016/j.celrep.2021.109836. PMC 8582597. PMID: 34686347.
- “Schedules of Controlled Substances: Placement of 2,5-dimethoxy-4-iodoamphetamine (DOI) and 2,5-dimethoxy-4-chloroamphetamine (DOC) in Schedule I.” Source: www.regulations.gov.
- “Statutes & Constitution: View Statutes: Online Sunshine.” Source: leg.state.fl.us.