The online market for research chemicals, including MDEA (3,4-methylenedioxy-N-ethylamphetamine), has increased in recent years, offering consumers a convenient platform to purchase these substances. However, the proliferation of online vendors selling MDEA and other designer drugs raises serious concerns.
One of the primary issues with MDEA research chemical sellers is the need for more regulation and oversight in this industry. These vendors often operate in a legal grey area, exploiting loopholes in drug control laws to offer substances for sale. As a result, more quality control and product consistency must be maintained, putting buyers at risk of receiving impure or contaminated substances. This poses significant health risks as the actual composition and safety of these research chemicals remain uncertain.
Furthermore, the online availability of MDEA and other designer drugs can appeal to individuals seeking to experiment with psychoactive substances, including those with little knowledge of their potential risks. This accessibility can lead to misuse and dangerous experimentation, potentially harming users’ physical and mental health.
Another concern is the lack of age verification or proper screening measures when purchasing these substances online. This means that minors and vulnerable individuals can easily access and buy these research chemicals, which can have devastating consequences.
Moreover, the marketing tactics employed by some MDEA research chemical sellers often border on the unethical. They may make unverified claims about the safety or effects of these substances, potentially misleading buyers and downplaying the associated risks.
Contents
- 1 Summary
- 2 History and culture
- 3 Chemistry
- 4 Pharmacology
- 5 Subjective effects
- 6 Toxicity
- 7 Legal status
- 8 FAQ
- 8.1 1. What is MDEA?
- 8.2 2. What are the effects of MDEA?
- 8.3 3. How is MDEA different from MDMA?
- 8.4 4. Is MDEA legal?
- 8.5 5. What are the risks associated with MDEA use?
- 8.6 6. Is MDEA addictive?
- 8.7 7. Can MDEA be combined with other substances?
- 8.8 8. How can I reduce harm while using MDEA?
- 8.9 9. Is MDEA being actively researched?
- 9 References
Summary
3,4-Methylenedioxy-N-ethylamphetamine, commonly referred to as MDEA or colloquially as Eve, belongs to the lesser-known entactogen category within the amphetamine class. Chemically akin to MDMA and MDA, MDEA exerts its effects by elevating serotonin, norepinephrine, and dopamine levels in the brain.
The earliest recorded human encounter with MDEA dates back to 1976 when Alexander Shulgin, a prominent chemist and researcher, noted its striking resemblance to MDMA in terms of effects and potency. However, he observed that MDEA acted more rapidly and had a shorter duration of action. The synthesis of MDEA and an array of related compounds and their pharmacological assessment was documented in 1980. MDEA was also featured in Shulgin’s 1991 publication “PiHKAL” (Phenethylamines I Have Known and Loved).
In the United States, MDEA emerged on the recreational scene in 1985 as a legal alternative to MDMA, which had recently been banned. However, just two years later, the authorities classified it as a Schedule I substance. Consequently, MDEA has seldom been available as a standalone substance and has predominantly been utilized as an occasional additive or substitute component in “Ecstasy” pills.
Minimal information is accessible regarding the pharmacological characteristics, metabolic processes, and toxicity associated with MDEA. Therefore, exercising great caution and employing harm reduction practices is highly recommended when considering this potentially habit-forming entactogenic substance.
| Identifiers | |
|---|---|
| show IUPAC name | |
| CAS Number | 82801-81-8 |
| PubChem CID | 105039 |
| ChemSpider | 94775 |
| UNII | ML1I4KK67B |
| ChEBI | CHEBI:132237 |
| CompTox Dashboard (EPA) | DTXSID70860971 |
| ECHA InfoCard | 100.231.031 |
| Chemical and physical data | |
| Formula | C12H17NO2 |
| Molar mass | 207.273 g·mol−1 |
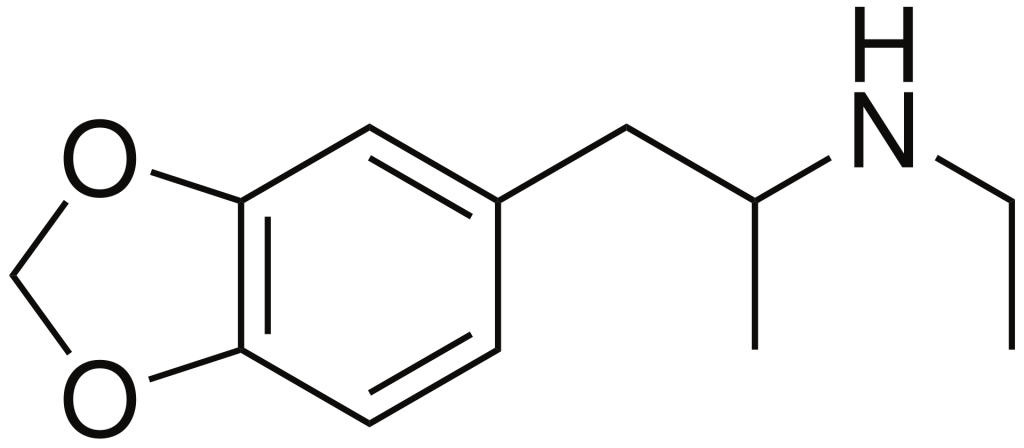
History and culture
In the United States, MDEA made its recreational debut in 1985, serving as a legal substitute for the recently banned MDMA. However, under the Federal Analogue Act, it was reclassified as a Schedule I substance on August 13, 1987. Since then, MDEA has seldom been available as a standalone product and has primarily served as an intermittent supplement or replacement component in “Ecstasy” pills. For instance, studies conducted in the 1990s revealed that MDEA was present in roughly four per cent of ecstasy tablets.
Although MDEA shares many of the fundamental entactogenic attributes of MDMA, it is marginally less potent and is often described as having a more “stoning” effect. It lacks the pro-social and energizing “magic” many party-goers seek in their MDMA experiences. Consequently, it is generally considered less desirable than MDMA by most individuals. As a result, MDEA is rarely produced and distributed in the illicit drug market, typically appearing only in limited quantities synthesized and disseminated by amateur clandestine chemists.
Chemistry
MDEA, scientifically known as 3,4-methylenedioxy-N-ethylamphetamine, belongs to the synthetic amphetamine chemical category. Compounds within the amphetamine class are characterized by a phenethylamine core, which consists of a phenyl ring linked to an amino (NH2) group via an ethyl chain. Additionally, there is an additional methyl substitution at Rα. Furthermore, MDEA features an ethyl substitution on RN, extending one carbon beyond the methyl group present in MDMA. Moreover, MDEA includes substitutions at R3 and R4 of the phenyl ring, where oxygen groups are integrated into a methylenedioxy ring through a methylene bridge. This methylenedioxy ring is a common feature shared by other entactogens such as MDMA, MDA, and MDAI.
Pharmacology
MDEA is a compound that both releases and inhibits the reuptake of monoamine neurotransmitters, specifically serotonin, dopamine, and noradrenaline, as research indicates. These neurotransmitters play crucial roles in regulating concentration, motivation, pleasure, and reward perception.
The “stoning” effects associated with MDEA usage are believed to be attributed to its relatively higher activity in releasing serotonin than dopamine, in contrast to MDMA.
Additionally, MDEA triggers the release of oxytocin and prolactin, two hormones presently under investigation for their potential involvement in modulating feelings of trust and love.
Subjective effects
Disclaimer: The following effects are derived from the Subjective Effect Index (SEI), a compilation of anecdotal user reports and personal analyses contributed by PsychonautWiki contributors. It is essential to approach these effects with a critical perspective.
Additionally, it is crucial to recognize that these effects may not manifest consistently or predictably. Higher doses may increase the likelihood of experiencing the complete range of impact. Conversely, higher doses can also lead to adverse effects, including addiction, severe injury, or even death ☠.
Physical:
- Stimulation & Sedation: MDEA is often noted for being less stimulating and invigorating than MDMA. Unlike MDMA, which energizes users for activities like dancing and running, MDEA offers a more subdued, mildly to moderately forced stimulation. Higher doses can lead to jaw clenching, involuntary body movements, and shaky sensations, resulting in reduced motor control, albeit to a lesser extent than MDMA.
- Spontaneous Bodily Sensations: MDEA produces a body high characterized by moderate to intense euphoria that envelops the entire body. This sensation can become overwhelmingly pleasurable at higher doses, even to the point of immobilizing the user. This feeling steadily intensifies with the onset, reaching its peak before gradually diminishing.
- Physical Euphoria: Users may experience a sense of physical well-being and pleasure.
- Perception of Bodily Heaviness: MDEA often induces a feeling of bodily weightiness, contributing to its “stoning” reputation compared to other entactogens that tend to have more pronounced stimulating effects.
- Tactile Enhancement: Users may notice heightened sensitivity to tactile sensations.
- Vibrating Vision: At high doses, users may experience rapid, involuntary eye movements (nystagmus), causing blurriness and temporary visual impairment.
- Temperature Regulation Suppression: MDEA can lead to a significant and sustained rise in core body temperature due to its serotonin-releasing properties. Caution is essential, as excessively high doses can result in serotonin syndrome, which can be life-threatening if left untreated.
- Increased Bodily Temperature: Similar to the above point, MDA’s serotonin-releasing effects often lead to a noticeable and consistent increase in body temperature.
- Dehydration: MDEA, like other entactogenic stimulants, may cause dry mouth and feelings of dehydration due to an elevated heart rate and metabolism. It is crucial to stay hydrated, but overconsumption of water can lead to water intoxication, so users should sip water cautiously.
- Difficulty Urinating: Higher doses of MDEA can make urination challenging temporarily. This effect is harmless and caused by MDEA’s promotion of anti-diuretic hormone release, which regulates urination. Placing a warm compress over the genital area can help alleviate this symptom.
- Increased Blood Pressure
- Increased Heart Rate
- Increased Perspiration
- Bodily Control Enhancement
- Appetite Suppression
- Brain Zaps
- Pupil Dilation
- Stamina Enhancement
- Teeth Grinding: Typically more noticeable at higher doses but to a lesser extent than associated with MDMA.
- Temporary Erectile Dysfunction
Visual:
Enhancements:
- Colour Enhancement
- Pattern Recognition Enhancement
Suppressions:
- Double Vision
- Distortions
- Tracers
- Symmetrical Texture Repetition
Hallucinatory States:
- MDEA can induce various low and high-level hallucinatory states, although these effects are less consistent and reproducible than traditional psychedelics. These hallucinatory states are often more prevalent during the comedown phase and may include:
- Peripheral Information Misinterpretation
Cognitive:
- Anxiety Suppression
- Empathy, Affection, and Sociability Enhancement: While present, these effects may not be as consistent, pronounced, consequential, or therapeutic as those experienced with MDMA. With repeated use, the empathic qualities may diminish.
- Disinhibition
- Cognitive Euphoria: Strong emotional euphoria and happiness, likely stemming from serotonin and dopamine release.
- Increased Music Appreciation
- Time Distortion: MDEA can create a strong sense of time compression, making time appear to pass more quickly.
- Compulsive Redosing
- Creativity Enhancement
- Focus Enhancement: Most effective at low to moderate doses; higher doses may impair concentration.
- Immersion Enhancement
- Motivation Enhancement
- Increased Libido
- Mindfulness
- Thought Acceleration
- Wakefulness
Auditory:
Enhancements
Hallucinations
Distortions
Transpersonal Effects:
- Existential Self-Realization
- Unity and Interconnectedness: Experiences of unity and interconnectedness are everyday within MDEA, particularly between levels 1 and 2.
After:
- The offset phase of a stimulant experience, commonly called a “comedown,” can be uncomfortable compared to the effects experienced during the peak. This discomfort often arises due to neurotransmitter depletion and may include:
- Anxiety
- Appetite Suppression
- Cognitive Fatigue
- Depression
- Dream Potentiation & Dream Suppression: Some users report vivid and occasionally unsettling dreams after consuming large doses of MDEA for several nights.
- Sleep Paralysis: Sleep paralysis may occur, especially with high doses or factors like sleep deprivation or physical fatigue.
- Irritability
- Motivation Suppression
- Thought Deceleration
- Wakefulness
Toxicity
Short-term physical health risks associated with MDEA consumption encompass dehydration, insomnia, hyperthermia, and hyponatremia. Engaging in continuous physical activity without adequate rest or rehydration can lead to dangerous increases in body temperature. The stimulatory and euphoric effects of the drug may cause users to be unaware of their energy expenditure for extended periods. The use of diuretics, such as alcohol, can exacerbate these risks.
The precise toxic dosage of MDEA remains unknown, but it is considered to be significantly higher than the dose required to produce its effects.
Neurotoxicity: MDMA’s neurotoxic potential, akin to MDMA, has been debated. Scientific consensus suggests that while MDEA can be used safely in a responsible context, repeated or high doses are likely to have neurotoxic and cardiotoxic effects to some degree.
MDEA, like other potent serotonin-releasing agents, is thought to down-regulate serotonin reuptake transporters in the brain. The brain’s rate of recovery from these serotonergic changes remains unclear. Some studies have shown lasting serotonergic changes in animals exposed to MDMA, which likely applies to MDEA as well. Other studies suggest the brain may recover from serotonergic damage.
Cardiotoxicity: Similar to MDMA, long-term heavy use of MDEA is likely cardiotoxic, potentially causing valvulopathy through its actions on the 5-HT2B receptor. One study reported that 28% of long-term MDMA users developed clinically evident valvular heart disease.
It is strongly recommended that harm reduction practices be employed when using this substance.
Tolerance and Addiction Potential:
MDEA, like other stimulants, can be moderately addictive with a high potential for abuse, leading to psychological dependence in some users. Individuals who become addicted may experience cravings and withdrawal effects upon discontinuation.
Tolerance to MDEA’s effects develops with prolonged use, necessitating larger doses to achieve the same impact. It takes approximately 1-1.5 months for patience to reduce by half and 2-3 months to return to baseline (without further consumption). MDEA exhibits cross-tolerance with all dopaminergic and serotonergic stimulants and entactogens, meaning that its use can reduce the effects of these substances.
Dangerous Interactions: Warning: Combining psychoactive substances can be hazardous, as some interactions can be life-threatening. Always conduct independent research to ensure the safety of combining two or more substances. Some known dangerous interactions include:
- 25x-NBOMe & 25x-NBOH: Combining these compounds with MDEA should be strictly avoided due to the risk of excessive stimulation, heart strain, increased blood pressure, panic attacks, thought loops, seizures, and, in extreme cases, heart failure.
- Alcohol: Combining alcohol with stimulants can be risky as stimulants can mask alcohol’s depressant effects, potentially leading to over-intoxication. After the motivation wears off, depressant effects may predominate, resulting in blackouts and severe respiratory depression.
- DXM: Combinations with DXM should be avoided due to its inhibitory effects on serotonin and norepinephrine reuptake, which can increase the risk of panic attacks, hypertensive crisis, or serotonin syndrome when combined with serotonin releasers like MDEA.
- MDMA: Combining MDEA with other stimulants can increase the risk of neurotoxicity, excessive blood pressure, and heart strain (cardiotoxicity).
- MXE: Some reports suggest that combining MXE with MDEA can dangerously elevate blood pressure and increase the risk of mania and psychosis.
- Dissociatives: Combining MDEA with dissociatives carries a risk of delusions, mania, and psychosis, which may be amplified when both are used together.
- Stimulants: Combining MDEA with other stimulants like cocaine can increase heart rate and blood pressure to dangerous levels.
- Tramadol: Tramadol lowers the seizure threshold, and combining it with stimulants like MDEA can increase this risk.
- MAOIs: Combining MDEA with MAOIs can raise neurotransmitter levels to dangerous or fatal, leading to potentially life-threatening conditions.
- Serotonin Syndrome Risk: MDEA use may lead to dangerously high serotonin levels when combined with substances like MAOIs, serotonin releasers (e.g., MDMA), SSRIs, SNRIs, and 5-HTP.
It is essential to exercise caution and conduct thorough research when considering the combination of substances.
Legal status
Internationally, MDEA is classified as a Schedule I substance under the Convention on Psychotropic Substances 1971. Here is its legal status in select countries:
- Austria: MDEA is illegal to possess, produce, and sell, regulated under the SMG (Suchtmittelgesetz Österreich).
- Brazil: MDEA is prohibited for possession, production, and sale, listed on Portaria SVS/MS nº 344 as “MDE”.
- Canada: MDA is categorized in Schedule I under the CSDA.
- Germany: MDEA has been controlled under Anlage I BtMG (Narcotics Act, Schedule I) since April 15, 1991. Manufacturing, possessing, importing, exporting, buying, selling, procuring, or dispensing it without a license is illegal.
- Switzerland: MDEA is a controlled substance listed explicitly under Verzeichnis D.
- United Kingdom: MDEA is classified as a Class A drug.
- United States: MDEA is categorized as a Schedule I drug.
FAQ
1. What is MDEA?
MDEA, or “Eve,” is a synthetic substance from the substituted amphetamine chemical class. It shares structural similarities with MDMA and MDA and is known for its entactogenic effects.
2. What are the effects of MDEA?
MDEA produces results by increasing serotonin, norepinephrine, and dopamine levels in the brain. Its products include feelings of euphoria, increased sociability, empathy, and altered sensory perception. It can also lead to physical sensations such as stimulation, relaxation, and increased body temperature.
3. How is MDEA different from MDMA?
While MDEA shares some similarities with MDMA, it is often described as less stimulating and more “stoning.” It lacks the intense pro-socializing and energizing effects of MDMA, making it less popular among some users. It may also have a shorter duration of action.
4. Is MDEA legal?
The legal status of MDEA varies by country. It is often classified as a controlled or illegal substance in many places. Always check your local laws and regulations regarding MDEA before considering its use.
5. What are the risks associated with MDEA use?
Short-term risks include dehydration, insomnia, hyperthermia, and hyponatremia. Continuous physical activity without proper rest or hydration can be hazardous. Long-term use may have neurotoxic and cardiotoxic effects.
6. Is MDEA addictive?
MDEA has the potential for psychological dependence and addiction, especially with chronic use. Users may experience cravings and withdrawal effects upon discontinuation.
7. Can MDEA be combined with other substances?
Combining MDEA with other substances, especially stimulants or MAOIs (Monoamine Oxidase Inhibitors), can be dangerous and lead to adverse reactions or serotonin syndrome. Always research potential interactions and exercise caution.
8. How can I reduce harm while using MDEA?
Harm reduction practices include staying hydrated, taking breaks from physical activity, avoiding excessive alcohol consumption, and researching potential drug interactions. Starting with lower doses and using them in a safe and controlled environment is essential.
9. Is MDEA being actively researched?
MDEA has limited research compared to other substances, and its use is primarily associated with recreational purposes. Research on its pharmacological properties, safety, and potential therapeutic applications is limited.
References
- Explore “Read #22 2C-C | PiHKAL · info” on isomerdesign.com.
- Delve into the Neuropsychopharmacology and Toxicology of 3,4-methylenedioxy-N-ethyl-amphetamine (MDEA) with insights from Freudenmann, R. W., and Spitzer, M. (2004) in CNS drug reviews. The article can be found in Volume 10, Issue 2, pages 89–116. DOI: 10.1111/j.1527-3458.2004.tb00007.x, ISSN: 1080-563X.
- Discover Alexander Shulgin’s “Pharmacology Lab Notes #2” from 1976-1980 in Lafayette, CA. You can access this resource on Erowid.org at p206: Shulgin Lab Notes #2.
- Explore the work of Braun, U., Shulgin, A. T., and Braun, G. (February 1980) on centrally active N-substituted analogues of 3,4-methylenedioxyphenylisopropylamine (3,4-methylenedioxyamphetamine) in the Journal of Pharmaceutical Sciences, Volume 69, Issue 2, pages 192–195. DOI: 10.1002/jps.2600690220, ISSN: 0022-3549.
- Investigate “Read #106 MDE – PiHKAL · info.”
- Dive into the realm of Healing with entactogens through the perspectives of therapist and patient experiences in MDMA-assisted group psychotherapy, as documented by Passie, T. (2012). This work is affiliated with the Multidisciplinary Association for Psychedelic Studies (MAPS) and can be found under ISBN 9780979862274.
- Explore the phenomenon of drug-induced hyperthermia through the research of Nimmo, S. M., Kennedy, B. W., Tullett, W. M., Blyth, A. S., and Dougall, J. R. (October 1993) in Anaesthesia, Volume 48, Issue 10, pages 892–895. DOI: 10.1111/j.1365-2044.1993.tb07423.x, ISSN: 0003-2409.
- Investigate the impact of small ambient temperature changes on 3,4-methylenedioxymethamphetamine (MDMA)-induced serotonin neurotoxicity and core body temperature in rats, as studied by Malberg, J. E., and Seiden, L. S. (July 1 1998) in The Journal of Neuroscience, Volume 18, Issue 13, pages 5086–5094. ISSN: 0270-6474.
- Explore the secretion of vasopressin and oxytocin in response to ecstasy consumption in a clubbing population as reported by Wolff, K., Tsapakis, E. M., Winstock, A. R., Hartley, D., Holt, D., Forsling, M. L., and Aitchison, K. J. (May 2006) in the Journal of Psychopharmacology, Volume 20, Issue 3, pages 400–410. DOI: 10.1177/0269881106061514, ISSN: 0269-8811.
- Investigate the reorganization of ascending 5-HT axon projections in animals previously exposed to the recreational drug (+/-)3,4-methylenedioxymethamphetamine (MDMA, “ecstasy”) as described by Fischer, C., Hatzidimitriou, G., Wlos, J., Katz, J., and Ricaurte, G. (August 1995) in The Journal of Neuroscience, Volume 15, Issue 8, pages 5476–5485. ISSN: 0270-6474.
- Discover the in vivo detection of short- and long-term MDMA neurotoxicity through a positron emission tomography study in the living baboon brain as presented by Scheffel, U., Szabo, Z., Mathews, W. B., Finley, P. A., Dannals, R. F., Ravert, H. T., Szabo, K., Yuan, J., and Ricaurte, G. A. (June 1998) in Synapse (New York, N.Y.), Volume 29, Issue 2, pages 183–192. DOI: 10.1002/(SICI)1098-2396(199806)29:2<183::AID-SYN9>3.0.CO;2-3, ISSN: 0887-4476.
- Explore preliminary findings on cortical serotonin transporter density and verbal memory in individuals who stopped using 3,4-Methylenedioxymethamphetamine (MDMA or “Ecstasy”) by Reneman, L., Lavalaye, J., Schmand, B., Wolff, F. A. de, Brink, W. van den, Heeten, G. J. den, and Booij, J. (October 1 2001) in Archives of General Psychiatry, Volume 58, Issue 10, page 901. DOI: 10.1001/archaic.58.10.901, ISSN: 0003-990X.
- Investigate brain serotonin transporter binding in former users of MDMA (‘ecstasy’) through the research of Selvaraj, S., Hoshi, R., Bhagwagar, Z., Murthy, N. V., Hinz, R., Cowen, P., Curran, H. V., and Grasby, P. (April 2009) in The British Journal of Psychiatry: The Journal of Mental Science, Volume 194, Issue 4, pages 355–359. DOI: 10.1192/bjp.bp.108.050344, ISSN: 1472-1465.
- Examine the phenomenon of drug-induced Valvulopathy with an update from Elangbam, C. S. (October 2010) in Toxicologic Pathology, Volume 38, Issue 6, pages 837–848. DOI: 10.1177/0192623310378027, ISSN: 0192-6233.
- Investigate the possible association between 3,4-methylenedioxymethamphetamine abuse and valvular heart disease, as discussed by Droogmans, S., Cosyns, B., D’haenen, H., Creeten, E., Weytjens, C., Franken, P. R., Scott, B., Schoors, D., Kemdem, A., Close, L., Vandenbossche, J.-L., Bechet, S., and Van Camp, G. (November 1 2007) in The American Journal of Cardiology, Volume 100, Issue 9, pages 1442–1445. DOI: 10.1016/j.amjcard.2007.06.045, ISSN: 0002-9149.
- Explore the dose-independent occurrence of seizures with tramadol, as documented by Talaie, H., Panahandeh, R., Fayaznouri, M. R., Asadi, Z., and Abdollahi, M. (2009) in the Journal of Medical Toxicology, Volume 5, Issue 2, pages 63–67. DOI: 10.1007/BF03161089, eISSN: 1937-6995, ISSN: 1556-9039.
- Delve into the interactions between monoamine oxidase inhibitors, opioid analgesics, and serotonin toxicity, as Gillman, P. K. (2005) explored in the British Journal of Anaesthesia, Volume 95, Issue 4, pages 434–441. DOI: 10.1093/bja/aei210, eISSN: 1471-6771, ISSN: 0007-0912. PMID: 16051647.
- Gain insights into the regulatory landscape with the “CONVENTION ON PSYCHOTROPIC SUBSTANCES 1971” (PDF) from the United Nations. Accessed on December 16, 2019.
- Understand the regulations governing psychotropic substances through the “Controlled Drugs and Substances Act – SCHEDULE I” provided by the Government of Canada. Retrieved on December 16, 2019.
- Explore the “Dritte Verordnung zur Änderung betäubungsmittelrechtlicher Vorschriften” (in German) from Bundesanzeiger Verlag. Retrieved on December 15, 2019.
- Investigate “Anlage I BtMG” (in German) from Bundesministerium der Justiz und für Verbraucherschutz. Retrieved on December 15, 2019.
- Explore “§ 29 BtMG” (in German) from Bundesministerium der Justiz und für Verbraucherschutz. Retrieved on December 15, 2019.
- Understand the regulatory framework with “Verordnung des EDI über die Verzeichnisse der Betäubungsmittel, psychotropen Stoffe, Vorläuferstoffe und Hilfschemikalien” (in German) from Bundeskanzlei [Federal Chancellery of Switzerland]. Retrieved on January 1, 2020.