Beautiful Plants For Your Interior
Summary
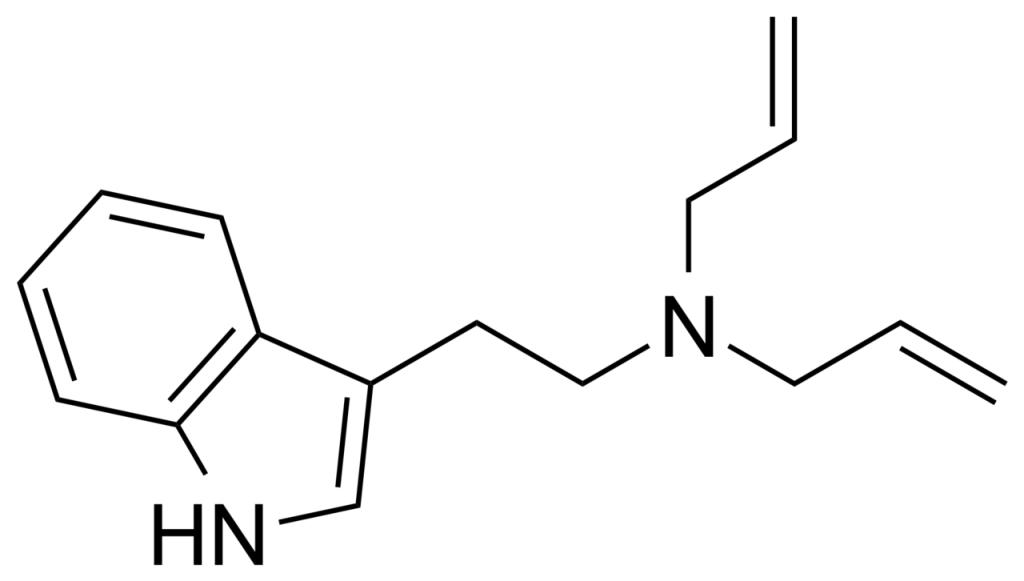
N, N-Diallyltryptamine, often referred to as DALT, is a derivative of tryptamine that has emerged as a novel psychoactive substance. It is known for its use as an intermediate compound in the synthesis of radiolabeled diethyltryptamine.
One of the notable effects associated with DALT is a significant increase in body temperature, which bears resemblance to the effects of 2,4-Dinitrophenol (DNP). Unlike DNP, DALT is considered to be a safer option due to its affiliation with the tryptamine family. DALT exhibits relatively mild psychoactive properties, particularly when compared to other tryptamines. The impact of this substance on body temperature and its low toxicity profile has prompted consideration of its potential application in emergencies within cold weather environments or as a supplement for weight loss.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 60676-77-9 |
|---|---|
| PubChem CID | 24839550 |
| ChemSpider | 21250454 |
| UNII | 85113MO9BC |
| Chemical and physical data | |
| Formula | C16H20N2 |
| Molar mass | 240.350 g·mol−1 |
FAQ
1. What is DALT?
DALT, or N, N-Diallyltryptamine, is a chemical compound classified as a derivative of tryptamine. It is known for its association with the tryptamine family and has been recognized as a novel psychoactive substance.
2. What is the primary use of DALT?
DALT has been employed as an intermediate compound in the preparation of radiolabeled diethyltryptamine. This application is a part of scientific research and experimentation.
3. What are the notable effects of DALT?
One of the more distinctive effects associated with DALT is a significant increase in body temperature. This effect is reminiscent of the temperature-related effects of 2,4-Dinitrophenol (DNP).
4. Is DALT considered safe to use in comparison to other substances like DNP?
DALT is a safer option compared to substances like DNP. This is attributed to its affiliation with the tryptamine family. However, it’s important to note that various factors can influence the safety of any substance, and responsible use is crucial.
5. How psychoactive is DALT in comparison to other tryptamines?
DALT is considered to have relatively weak psychoactive properties, especially when contrasted with other members of the tryptamine class.
6. Are there potential alternative applications for DALT?
Due to its effects on body temperature and its low toxicity profile, DALT has sparked interest as a potential option for emergencies in cold-weather environments. It has also been considered for use as a weight loss supplement.
7. Is DALT legal for recreational use, and where can it be found?
The legal status of DALT can vary by country and region. It’s essential to be aware of and abide by local laws and regulations regarding its use and distribution.
8. Can DALT be used for therapeutic purposes?
The therapeutic potential of DALT has not been extensively studied. As with any psychoactive substance, it should only be considered for therapeutic use under the guidance of qualified medical professionals in clinical settings.
9. Where can I find more information about DALT?
To access further information about DALT, consider consulting scientific literature, academic research, or engaging with experts in the field of psychoactive substances. Always prioritize safety and responsible use when considering the consumption of any such compounds.
References
- In October 2015, Michely JA, Helfer AG, Brandt SD, Meyer MR, and Maurer HH conducted a study on the metabolism of the new psychoactive substances N,N-diallyltryptamine (DALT) and 5-methoxy-DALT. This research explored their detectability in urine using various analytical techniques, including GC-MS, LC-MSn, and LC-HR-MS-MS. The findings were published in “Analytical and Bioanalytical Chemistry” (Volume 407, Issue 25), with a DOI of doi:10.1007/s00216-015-8955-0. The study is indexed under PMID 26297461.
- In February 2017, Michely JA, Brandt SD, Meyer MR, and Maurer HH continued their research in the same area. This study focused on the detectability of psychoactive substances in biological samples, utilizing analytical methods including LC-MS/MS. The results were published in “Analytical and Bioanalytical Chemistry” (Volume 409, Issue 6) and can be accessed via the DOI doi:10.1007/s00216-016-0117-5. The study is referenced under PMID 27933361.
- In 2008, Brandt SD, Tirunarayanapuram SS, Freeman S, Dempster N, Barker SA, Daley PF, and others engaged in the microwave-accelerated synthesis of deuterated N,N-dialkylated-[α,α,β,β-d4]-tryptamines. This research was published in the “Journal of Labelled Compounds and Radiopharmaceuticals” (Volume 51, Issue 14) with a DOI of doi:10.1002/jlcr.1557.